Novel magnets made from the strongest known hydrogen bond
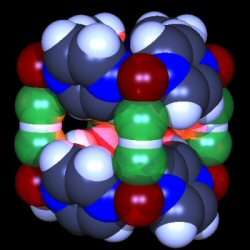
A team of scientists from the US, the UK and Germany has been the first to make a magnetic material constructed from nature's strongest known hydrogen bond. Hydrogen bonds are responsible for many of the properties of water and for holding together the DNA double helix.
However, hydrogen bonds are normally rather weak but the new compound contains the bifluoride ion in which a hydrogen atom is tightly bound to two fluorine atoms. This leads to the new magnet being stable up to 200 degrees Celsius.
The work is published in the latest issue of the journal Chemical Communications, where it features on its cover.
The magnetic properties of the material were measured using muons by a team at Oxford University, headed by Stephen Blundell, Professorial Fellow in Physics at Mansfield College. He said: ‘Muons are tiny, sub-atomic particles which can be implanted into materials. They behave like tiny gyroscopes and spin round when they experience a magnetic field.’
Using this method, which is uniquely sensitive to magnetism in these types of magnetic material, the researchers found that there is no overall magnetism at room temperature; however, as the temperature is lowered, copper magnetic moments begin to align, producing a microscopic magnetic field visible to the implanted muons below a temperature of 1.54 Kelvin.
These experiments were performed at ISIS, the world's most intense source of pulsed muons, located in Oxfordshire, UK.
The team hopes that the magnetic studies will help them understand to what extent bifluoride units and their hydrogen bonds influence the spin arrangement on neighbouring magnetic centres.
Source: University of Oxford
















