Self-assembling and biologically active artificial pores may lead to a future full of holes
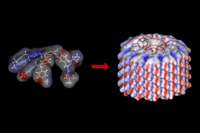
In the August 12 issue of the journal Nature, researchers at the University of Pennsylvania detail the creation of a library of small protein-like molecules that can self-assemble to form hollow corkscrew-like pores that could mimic pores seen in living systems. These molecules, formed from short chains of amino acids called peptides attached to tree-like fragments called dendrons, represent the first successful attempt at creating man-made pores that can form in solution and in bulk.
"Using variations on naturally-occurring and synthetic amino acids, we could create a nearly unlimited variety of pores of different shapes and sizes," said Virgil Percec, a professor in Penn's Department of Chemistry. "We can foresee creating pores to mimic biological function, to filter out one molecule from a solution or to carry specific molecules across cellular membranes."
In nature, proteins that form hollow pores are ubiquitous to life, performing many essential tasks such as forming channels to cross cell membranes, generating chemical energy, guiding the shape of newly-made proteins and even puncturing holes in the cell walls of bacteria.
"The application of the technology could, for example, lead to better means of filtering drinking water from seawater or to an entirely new class of antibiotics by creating pores that poke holes in harmful bacteria," Percec said.
Each peptide subunit has arm-like projections that allow it to bind to similar peptides in a spiraling fashion. Held together by hydrogen bonds, the stable helix created by these peptides forms in such a way as to create a tube or channel, the width of which can be modified by using different combinations of amino acids.
"We found that the ability of the peptides to recognize each other and self-assemble is sufficiently robust to tolerate a wide range of modifications yet still remain capable of forming pores," Percec said.
According to the researchers, the self-assembling peptides can form in and on the surface of microbial cell membranes, a breakthrough with enormous therapeutic potential. The ability to mimic the function of natural pores has long been a goal made very difficult by the complex chemistry of proteins. Indeed, life as we know it would not exist if it were not for the membranes that separate cells from the outside world. Likewise, cells would not exist without the protein pores to cross these membranes, importing substances necessary to sustain the cell and exporting wastes or products needed by other cells.
"It has come to our attention that, if we cannot precisely recreate the structure of proteins found in nature, then perhaps we can mimic their function and create new biologically inspired systems that achieve the same result," Percec said. "We can also imagine a great many non-biological systems and chemical processes that could benefit from a few good holes."
Funding for the research was provided by the National Science Foundation and the Office of Naval Research.
Penn researchers involved in the study include Andres E. Dulcey, Venkatachalapathy S. K. Balagurusamy, Yoshiko Miura, Jan Smidrkal, Mihai Petereca, Sami Nummelin and Ulrica Edland of the Department of Chemistry; Sergei A. Vinogradov of the Department of Biochemistry and Biophysics; and Paul A. Heiney of the Department of Physics and Astronomy.
Source: University of Pennsylvania
















