New mass-spectrometer analyzes proteins faster and at a lower cost
The sequencing of the human genome was only the beginning of a much more complex task – deciphering the secrets of cellular chemistry and the mechanisms of disease. While the genome serves as a blueprint to understanding the body, proteins represent the materials that carry out these plans.
Researchers at the Georgia Institute of Technology have developed a device that has the potential to significantly reduce the time needed to analyze these important proteins, shortening development time for new drugs and bringing down the overall cost of protein analysis technology. According to findings published in Applied Physics Letters, the device can potentially analyze proteins much faster, more gently and at a lower cost.
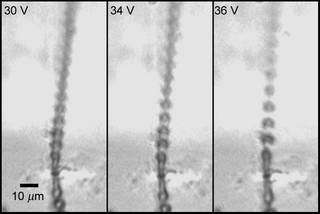
A stroboscopic image of a jet of droplets generated by AMUSE at a rate of more than a million droplets per second. An ability to produce very fine (only a few micrometers in diameter) charged droplets is critical to efficient mass spectrometric analysis.
There are about 2 million distinct proteins in the human body. That’s a lot of proteins – and the future of personalized medicine depends on a better understanding of proteins, including their structure and interactions with drugs and medical devices.
“The device has the potential to completely change the landscape of this field,” said Andrei Fedorov, an associate professor in the Woodruff School of Mechanical Engineering at Georgia Tech who leads the project. Fedorov’s collaborators on the project include Professor F.L. Degertekin from the Woodruff School of Mechanical Engineering and Professor F.M. Fernandez from the School of Chemistry and Biochemistry.
The device is a critical component of a mass spectrometer, an instrument that can detect proteins present even in ultra-small concentrations by measuring the relative masses of ionized atoms and molecules. Mass spectrometers can provide a complete protein profile and essentially make proteomics, the study of how proteins are produced and interact within an organ, cell or tissue, possible.
“You need to be able to take a blood sample, pass it through a system and figure out the complete protein profile of the human plasma. It’s an extremely technology-intensive process and you need to have a technology to do this kind of testing quickly and inexpensively,” Fedorov said.
But before the mass spectrometer can analyze a sample, molecules must first be converted to gas-phase charged ions through electrospray ionization (ESI), a process that produces ions by evaporating charged droplets obtained through spraying or bubbling.
Georgia Tech’s AMUSE (Array of Micromachined Ultra Sonic Electrospray) technology has several key advantages over currently available electrospray methods. In AMUSE, the sample aerosolization and protein charging processes are separated, giving AMUSE the unique ability to operate at low voltages with a wide range of solvents. In addition, AMUSE is a nanoscale ion source and drastically lowers the required sample size by improving sample use.
Also important, AMUSE is a “high-throughput” microarray device, meaning that it can analyze many more samples at a time than a conventional electrospray device.
This innovation will be particularly useful for the pharmaceutical industry. Drugs target certain proteins to achieve their designed effect on the body. The pharmaceutical industry must test large numbers compounds on even larger numbers of proteins to determine what effect a substance has on the body and whether or not it is safe. With AMUSE, the time-consuming process could be streamlined considerably, which could significantly shorten drug development time.
In addition to its ability to handle a much higher number of samples, AMUSE can also be manufactured more cheaply than current ESI devices. Conventional electrospray devices in mass spectrometers generally cost around $150 a piece and must be cleaned after each sample is analyzed. AMUSE could be made disposable and mass produced at a few dollars a piece, making Georgia Tech’s device a key step toward more affordable mass spectrometers for clinical applications.
For example, to determine whether a patient has cancer, a small blood sample is typically frozen and sent out to a testing lab at another facility. This freezing process and trip to the lab have a significant impact, damaging the proteins and possibly giving an incomplete analysis. In the future, with a powerful and portable mass spectrometer, it may be possible for a doctor to take a sample directly from the patient, place it in the device and receive an analysis on the spot.
Source: Georgia Institute of Technology


















