Don't stress! Bacterial crisis command center revealed
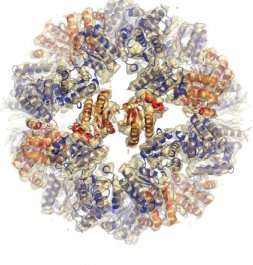
A bacteria cell's 'crisis command centre' has been observed for the first time swinging into action to protect the cell from external stress and danger, according to new research out today (3 October) in Science.
The research team behind today's study says that finding out exactly how bacteria respond and adapt to stresses and dangers is important because it will further their understanding of the basic survival mechanisms of some of the most resilient, hardy organisms on Earth.
The crisis command centre in certain bacteria cells is a large molecule, dubbed a 'stressosome' by the scientists behind today's research. These cells have around 20 stressosomes floating around inside them, and although scientists knew they played an important role in the cell's response to stressful situations, the complexities of this process had not been fully understood until now.
If a bacteria cell finds itself in a dangerous situation - for example, if the temperature or saltiness of the bacteria's environment reach dangerous levels which threaten the survival of the bacteria -a warning signal from the cell's surface is transmitted into the cell.
Using cutting edge electron microscopy imaging techniques the authors of the new research observed that the stressosomes receive this warning signal, and in response several proteins called RSBT break away from the large stressosome. This breakaway triggers a cascade of signals within the cell which results in over 150 proteins being produced - proteins which enable the cell to adapt, react and survive in its new environment.
Professor Marin van Heel from Imperial College London's Department of Life Sciences, one of the corresponding authors of the study, explains: "The cascade of events inside bacteria cells that occurs as a result of stressosomes receiving warning signals leads to particular genes inside the cell being transcribed more. This means that some genes already active inside the cell are 'turned up' so that levels of particular proteins in the cell increase. These changes to the protein make-up of the cell enable it to survive in a hostile or challenging environment."
Dr Jon Marles-Wright from Newcastle University says: "Our work shows that cells respond to signals much like a dimmer on a light switch. Now we'll be building on this to work out how nature controls that dimmer switch. We wouldn't have been able to carry out this work without access to the Diamond synchrotron Light Source which has enabled us to examine the structures of individual stressosome proteins at atomic resolution."
Dr Tim Grant, one of Imperial's post doctoral researchers, adds that the key to bacteria cells' success at surviving in rapidly changing environments is their speedy response: "The cell's stressosomes are very good at their job as crisis command centres because they provide a very fast effective response to danger. The chain reaction they kickstart produces results really quickly which enables bacteria to adapt to changes in their surroundings almost instantaneously."
The team is now planning to collect very high resolution data of the stressosome complex on the world's newest high-resolution cryo electron microscope, the FEI "KRIOS" that has just been installed in the Max Planck Institute in Martinsried, Germany. Improving the resolution of the stressosome structure by a factor of two will lead to a resolution range normally only attainable by X-ray crystallography and will allow the researchers to directly see the amino-acid components of this fascinating complex.
Source: Imperial College London


















