No helicopter moms among Rutgers mutant mice
First, he discovered a gene that controls innate fear in animals. Now Rutgers geneticist Gleb Shumyatsky has shown that the same gene promotes "helicopter mom" behavior in mice. The gene, known as stathmin or oncoprotein 18, motivates female animals to protect newborn pups and interact cautiously with unknown peers.
This "fear gene" is highly concentrated in the amygdala, a key region of the brain that deals with fear and anxiety. Shumyatsky's newest finding could enhance our understanding of human anxiety, including partpartum depression and borderline personality disorders.
Shumyatsky is an assistant professor of genetics in the School of Arts and Sciences at Rutgers, The State University of New Jersey.
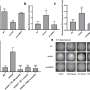
Working with female mice genetically engineered to have an inactive stathmin gene, Shumyatsky demonstrated that these mutant mice were slow to retrieve pups placed outside the nest at corners of the cage. Females with normally active stathmin, however, were quick to bring similarly dispersed pups back to the nest. In another experiment, knockout mice chose to rebuild nests in more vulnerable open spaces instead of in safe corners, where normal mice typically build nests.
The abnormal behavior, concludes Shumyatsky, is based on the mouse's lack of fear – in this case, fear for the safety of pups in her care. Retrieving wayward pups is a behavior motivated by innate fear of attack by predators, a likely outcome for wild pups that stray from the relative safety of a nest.
"The human analog might be parents on a playground with their children when it starts to thunder," said Shumyatsky. "The typical parental behavior would be to gather their children and seek shelter. Parents who behave as these mice do would say, 'so they get a little wet, what's the problem'. That's definitely not the kind of helicopter parenting that newborn mice need to survive, and by extension, the species needs to survive."
Shumyatsky and postdoctoral research collaborators Guillaume Martel and Akinori Nishi published their findings this week in the Proceedings of the National Academy of Sciences (PNAS) Early Edition online, and will be published in an upcoming issue of the PNAS print edition.
He contrasts the new findings with his earlier "fear gene" discovery, which focused solely on personal risk. "We now show that having less fear can profoundly change important behaviors for the survival of progeny and the species," he said.
The researchers conducted several experiments to rule out other reasons why the mutant mice were slow to retrieve pups. They ruled out diminished olfactory perception, as both normal and mutant mice located missing pups with equal speed using their sense of smell.
Both types of mice also equally passed object perception tests. The researchers then ruled out non-fear motivation, as both types stockpiled food with the expected urgency.
The one experiment that clinched fear-based motivation as the factor was when they "reminded" mice of their parental responsibility by first putting pups in the nest for five minutes before dispersing them. In this case, the mutant mouse retrieved pups as quickly as the normal mouse. This ruled out the possibility that mutant mice didn't retrieve pups because they lacked a fundamental knowledge of how to do it.
In the social behavior experiment, the mutant mice showed much less cautious behavior to other peers than did the normal mice. "The equivalent human behavior would be if a person hugged every stranger she met," said Shumyatsky. "In fact, that's something that humans with amygdalar damage might do – they're very trusting."
The research adds further evidence to the amygdala's role in controlling innate fears – specifically, the basolateral complex (BLA) of the amygdala. The BLA's role in forming memories for learned fear has been well-established, but its role in regulating innate behaviors has been less well understood.
Source: Rutgers University