Fruit flies all aglow light the way to cancer prevention
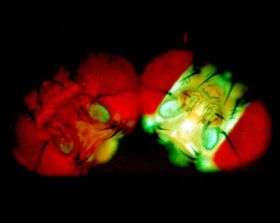
A green glow from a fruit fly is giving researchers the green light when they are on the right path in their quest to develop compounds that help prevent cancer.
The glow, the result of some tinkering in Drosophila, the workhorse of the genetics world, lets researchers know when powerful cancer-prevention signals similar to those spurred by protective chemicals in broccoli, cabbage, and other foods, have been turned on in the organism.
The chemical signaling system is one of the major ways that the body defends itself against toxic assaults and threats like cigarette smoke, diesel exhaust, and dangerous microbes. A gene known as KEAP1 senses danger and then unleashes NRF2, which triggers rampant anti-oxidant activity in a cell.
Now scientists from the University of Rochester Medical Center have discovered that the pathway, long recognized in people and other animals, is active in fruit flies, too, opening the door to faster, less expensive ways to find compounds that spur our natural anti-oxidant activity. The work, funded by the National Cancer Institute, is reported in the Jan. 15 issue of Developmental Cell.
“This is one of the main mechanisms the body uses to fight off the things that give you cancer,” said Dirk Bohmann, Ph.D., professor in the Department of Biomedical Genetics and a geneticist who studies fruit flies in an effort ultimately aimed at improving human health.
“This puts cells into an anti-oxidant defense mode. Drug development and testing is very, very expensive and time-consuming. This work should speed the development of new drugs aimed at preventing cancer,” added Bohmann.
Bohmann did the work along with former postdoctoral Gerasimos P. Sykiotis, M.D., Ph.D., who teamed up with Bohmann to develop novel approaches for the study of the NRF2 pathway after earning his medical and doctoral degrees from the University of Patras in Greece. Sykiotis is now with the Model Organisms Unit of the Novartis Institutes for Biomedical Research in Cambridge, Mass., where he is applying the genetic tools generated in the study to characterize the role of NRF2 signaling in Drosophila models of human diseases.
Scientists have known that the pathway exists in people, rodents, and zebrafish, and so Bohmann and Sykiotis went hunting for it in the fruit fly genome. They found that one form of a gene called CNC, which is widely known to be involved in determining the development of a fruit fly’s head, serves like NRF2, turning on cellular defenses on a broad scale.
The defenses include activation of molecules known as thioredoxins and glutathione S-transferases, which are anti-oxidants that help a cell get rid of toxins and damaged molecules in its environment. Unlike popular anti-oxidants in certain foods and vitamins, whose effects in the body are transient, Bohmann points out that a fundamental genetic change like a boost in NRF2 activity throughout an organism would supply an ongoing amplified anti-oxidant response.
While the main application of the work is in boosting the body’s ability to resist cancer, the research could also make a difference for patients who have cancer that is resistant to current drugs. In 2006, a team from Johns Hopkins showed how this same signaling pathway allows some cancer cells to fight off drugs intended to kill them. Gaining a foothold on the system in fruit flies gives researchers an added tool as they search for ways to thwart these rogue cancer cells.
In their experiments, Bohmann and Sykiotis modified fruit flies so they would glow green when exposed to ultraviolet light when the signaling pathway is functioning. Sure enough, flies with more active CNC glow more brightly than regular flies, giving the team an easy, visual way to see whether the pathway is activated.
The team demonstrated the technology using a compound called oltipraz, which targets the pathway and has been tested in people as a cancer-prevention agent. The flies that ate food with the compound glowed more strongly, demonstrating that the NRF2 pathway was more active in these flies.
“Turning on our natural anti-oxidants is big business for many companies trying to develop compounds to protect us from cancer and to slow the aging process,” said Bohmann. “The same genetic principles govern many organisms, from flies to rodents to people, and we’re hopeful that our tool in fruit flies will speed this work for the benefit of patients.”
When Bohmann and Sykiotis boosted the activity of the pathway, fruit flies were three times more likely to survive an exposure to a toxin than regular flies. And flies with a more active signaling system can live 10 percent longer than the other flies.
It’s the first time that the system, long known to be an important anti-oxidant and cancer prevention pathway, has also been shown to play a role in giving an organism a longer lifespan. The link gives new insight into the well-established connection between aging and cancer risk.
Two of Bohmann’s colleagues at the University of Rochester Medical Center are also studying the NRF2 pathway. Steve Georas, M.D., professor of medicine and chief of the Division of Pulmonary and Critical Care, is looking at the role of NRF2 in people with asthma. And Irfan Rahman, Ph.D., associate professor of Environmental Medicine, has shown how NRF2 protects the lungs of smokers against the assault of cigarette smoke and other pollutants. He has shown that organisms in which NRF2 is weakened or absent have weak lungs and are much more prone to conditions like emphysema.
Source: University of Rochester


















