New cell death pathway involved in sperm development
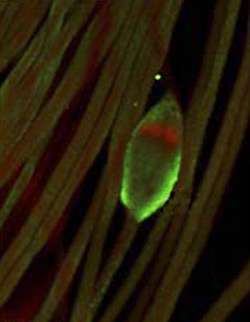
Heavy and bulky sperm would not be good swimmers. To trim down, sperm rely on cell death proteins called caspases, which facilitate the removal of unwanted cellular material and radically remodel these cells into their sleek, light shape. New research from scientists at the Howard Hughes Medical Institute and Rockefeller University has now uncovered a new pathway that regulates these killer proteins, yielding new knowledge about caspase function as well as insights into the causes of human infertility. The findings are reported this week in Public Library of Science Biology.
Cell death caspases, when activated, were thought to condemn a cell to certain death. But a few years ago Hermann Steller, head of the Laboratory of Cancer and Apoptosis Biology, and his colleagues discovered that caspases also function without entirely killing cells; instead, they are used to shape cells by dismantling unwanted bulk. “This process is very similar to apoptosis, or cell suicide,” explains Steller, who is Strang Professor at Rockefeller and an investigator at HHMI, “but in this case cells live.” And in Drosophila, when this cell death-like program goes awry, males become sterile.
Though quite a bit has been learned about how caspases are activated, very little is known about how unwanted caspase activity is restricted so that healthy, productive cells aren’t mistakenly target for death. So Steller and his colleagues wanted to figure out how caspases, which are expressed in all cells, are activated at the right time and at the right place; and in this case, how they do not kill off a cell entirely.
The researchers screened more than 1,000 sterile male fruitflies, looking for cellular differences between sterile flies and fertile ones. They then mapped these differences back to the genes to identify mutations along the Drosophila genome that made these fruitflies sterile. This process eventually pointed them to three distinct genes that encode different protein components of a complex called Cullin-3 ubiquitin ligase.
Cullins are members of the E3 ubiquitin ligase family, which label other proteins with ubiquitin, a molecule that marks them for degradation. It turns out that Cullin-3, in conjunction with two other proteins, activates caspases by degrading a caspase inhibitor. This, in turn, initiates a cell death-like program at the right time and at the right place — in the developing testes of Drosophila — and gets rid of unwanted cytoplasm and organelles. Before this study, only IAPs, another class of E3 ubiquitin ligases, had been identified as caspase regulators. Now, Steller and his group have found a new major player that regulates these killer proteins.
One of the proteins that form the Cullin-based complex in Drosophila has also been linked to male infertility in mice and humans. In mice, a mutation in the gene that encodes a protein called Klh110 causes male sterility. In humans, male infertility has been linked to this gene as well, although it is still not known whether this is due to the inability of Cullins to activate caspases and promote sperm differentiation.
The Steller lab initially focused on the role of Cullins during sperm development, but there is already data indicating that they also function to regulate caspases in somatic cells. It appears that cells use several different mechanisms simultaneously to protect themselves against unwanted caspase activity and death. This information provides new opportunities to develop drugs that can alter cell death for therapeutic purposes, either for cellular protection or cell killing — processes that range from neurodegenerative disease to cancer.
“Our findings provide a new framework to understand how apoptotic proteins are regulated for cellular remodeling.” says Steller. “And now, by having a more comprehensive picture of these different pathways and how they come together, we are prepared to look much more broadly at different cell death paradigms.”
Source: Rockefeller University















