February 1, 2006 feature
Nano this, Nano that, what the...
Nano has officially become the most misused word in the English language. Everything from the Ipod Nano to anything smaller than a Mac truck gets “nanoed” by clueless – or savvy, take your pick – marketing experts. It’s crept into everyday use as well: “I’ll be there in a nano.” Sure you will.
For the scientists who work with nanotech this must be frustrating indeed. A definition of nano is definitely in order. Nano is 10 to the power of -9. How small is that? A nanometer is one billionth of a meter. Small, indeed – in fact, so small it’s difficult to compare it to anything, only adding frustration to the roll of scientists trying to explain nanotechnology in laymen’s terms.
Let’s try anyway. A nanometer is 100,000 times smaller than the diameter of a human hair. There are as many nanometers in an inch as there are inches in 400 miles (25,344,000). About 3 to 6 atoms can fit inside a nanometer - depending on the atom size. Even when you see the comparisons, they mean nothing – it’s just too small - hence the attempt to make sense of it by tagging everything smaller than normal as “nano”.
Nanotechnology is more than just a hot theoretical topic now. Computer chips are now made using a 15 nanometer lithography process. But how can something so small be useful? After all, you can’t even see it under most microscopes. The answer lies in large numbers. A Pentium 4 Extreme Edition, for example, has 178 million transistors, all on the nanoscale – defined as smaller than 100 nanometers in length. This minuscule size allows processors to be faster, generate less heat and run using less energy.
At this scale, a material's properties change—things like electrical conductivity and mechanical strength are not the same as they are at micro size. With the recently developed atomic force microscope, nanoscientists have begun to manipulate matter on an atomic level to take advantage of these exciting new properties.
Their first attempts were childish – IBM made its name out of individual atoms, for example. But now nanotechnology is getting serious.
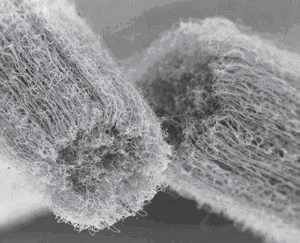
Consider the carbon at the nanoscale. Arrange carbon atoms one way you get a diamond – arranged another way, you get graphite. Nanoscientists have learned to arrange carbon atoms in a new way: tiny tubes about two nanometers in width that can look like chicken wire and can grow to a length many times their diameter. It's still the element carbon, but the carbon nanotube is a completely different material from either diamond or graphite – unlike any found in nature. These tubes may some day be the ultimate carbon fibers – theoretically, they are immensely strong.
Nanotechnology is not new. In fact, for over a century, chemists have developed the ability to arrange small numbers of atoms inside molecules – at a scale of less than 1.5 nanometers - leading to revolutions in drug design, plastics, and other areas.

Many of the processes we depend on, indeed, life itself, occur on the nanoscale. With the advent of nanotechnology, medical science has the potential ability to manipulate cells at the lowest level. Where they now use sledgehammer treatments – chemotherapy, for example, one day they may have a pair of needle-nosed pliers that could repair cells instead of just killing off the defective ones. As this article is being written, doctors are already using nanotech if the form of contrast agents for MRI scans. Gadolinium in DNA-sized carbon tubes boosts performance and reduces the metal’s high toxicity by containing the contrast agent and keeping it from affecting surrounding tissues.
Not all nanotech is based on carbon atoms and molecules. A new inorganic fullerene-like nanostructure – IF for short – is a material produced by the Weizmann Group. Instead of carbon, it’s made of tungsten disulfide. Unlike carbon based organic Fullerenes, IF is easier and less expensive to produce, is chemically stable, less reactive and less flammable. Organic Fullerenes are highly toxic while IF materials have been fully tested and deemed safe.
One of the most interesting new IF properties discovered is its extremely high degree of shock absorbing ability. The new Tungsten-based IF material has up to twice the strength of the best impact resistant materials currently used in protective armor such as boron carbide and silicon carbide. It’s over 5 times stronger than steel. Mixing IF with elastic materials could lead to new compounds which are both shock absorbing and flexible – perfect for ballistic armor.
Another important area for nanotechnology is coatings. New nanocoated metals have high wear resistance and resilience, better thermal shock resistance, resist fatigue, and have enhanced anti-microbial activity.
The list of potential uses is almost endless: In sports to make stiffer tennis racquets and sharper, more resistant snowboard edges, in lightweight nanometal foam helmets that provide up to 7 times the protection of conventional models. In medicine: nanocrystaline metals of silver and cobalt are effective antibacterial agents and can be used in air-conditioning systems to stop formation of bacterial slime. Nanocrystalline zinc oxide is used in sunscreens - zinc oxide and titanium oxide effectively absorb ultraviolet radiation. Industrial applications include: better lead acid batteries, a replacement for chrome, super lightweight parts and wear resistant coatings.
Fabricators produce nanocrystalline metals - alloys and composites - using a patented electrosynthesis process. They are able to make crystal sizes 1000-times smaller than those found in normal materials.
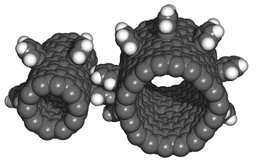
For scientists and engineers interested in exploring the properties of nanometals, they can be purchased on the Internet – get the research funding first because a 0.008” x 3” x 3” sample plate can set you back $2,000. Depending on the plate’s size and materials used, samples can cost as little as $115, however.
Nanomachines are not just on the drawing board anymore, a few working models– like the gears above – are regularly produced. Integrating them into useful systems is still a ways off, but the potential benefits are staggering.
Imagine submicroscopic machines that rove about the body and scrape plaque deposits from your artery walls, repair damage caused by trauma and attack cancer cells. Now that biotech is taking off as well, it should be interesting to see which approach – biotech or nanotech – offers the most benefit to medical science. In the end it may be a combination. Already a new field – Synthetic Biology – is exploring ways of duplicating biological processes with nanostructures.
Some futurists predict the advent of self-replicating nanomachines. These nanobots will be able to build copies of themselves and perform some useful service to humanity – or not. No discussion of nanotechnology can leave out the famous “grey goo” that represents trillions of self-replicating nanobots that literally eat the Earth to make more copies of themselves. While this science fiction plot is not probable, it should be studied carefully before unleashing a horde of hungry nanocreatures into the wild.
Closer to the truth are potential toxicity problems with fine nanomaterials. It’s not like you can filter them from the air – they are too small. Once escaped from the laboratory, they could become the Lilliputian Frankenstein of the future – forever blowing about in the wind forever changing anything they touch – like CFCs of steroids. All technologies have their problems and experts agree nanotechnology will be no exception.
But with all the money flowing into nanotech research, safety concerns will like as not be pushed aside to advancement science – much as they have in the past. At what price progress? With all the potential benefits nanotechnology has to offer, it’s sure to be a bargain.
by Philip Dunn, Copyright 2006 PhysOrg.com



















