Gene induces eyes in odd spots

A gene thought to play a relatively minor role in eye development is powerful enough on its own to initiate the formation of eyes in strange spots on a fruit fly's body, Indiana University Bloomington scientists have learned.
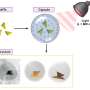
Biologist Justin Kumar and colleagues report in the March 15 issue of Developmental Biology that the gene sine oculis (or just "so") is capable of turning on a series of developmental cascades that result in the formation of retinas on traditionally non-retinal tissues including the antennae, legs, wings and even genitals.
"It may not seem like it, but this avenue of research really gets at the heart of the question: Why is eye development restricted to the head of flies, mice, and men?" Kumar said. "If you can break the system, you can learn a lot by comparing what's normal to what isn't."
SO is not the first Drosophila melanogaster protein discovered to be capable of initiating "ectopic" eyes in strange places. Quite the opposite is true, in fact. It has been shown that a number of nuclear proteins that form a regulatory network posses this unique property. It was believed that SO, a member of this network that also belongs to the SIX family of transcriptional regulators, was uniquely incapable of inducing retinal formation.
"Up until now it was thought that SO was the only factor within the retinal determination network that was unable to induce eye development," Kumar said. "So every paper -- including my own -- would cite this erroneous fact and try to build models around it. Our paper suggests SO is as 'powerful' as the other members of the cascade. Hopefully we can now draw simpler models of how these regulatory genes interact with each other and with other regulatory systems."
The finding also moves developmental biologists one step closer to understanding why, how and when eyes form in Drosophila and other animals and, perhaps just as importantly, why eye tissue normally forms on the head -- but no place else. Additionally, flies in which eye development genes are forcibly expressed in non-eye places help biologists like Kumar identify other genes that are involved in the formation of the retina.
"The SIX genes are expressed broadly, but you get eyes in only very specific locations," Kumar said. "There are clearly regulatory mechanisms that are stopping these genes from behaving inappropriately."
Kumar and colleagues employed a series of genetic tools to express SO in regions of Drosophila that normally doesn't express this factor. In all, the scientists attempted to induce eyes in 219 different developmental patterns. The scientists found the expression of SO was enough to induce eye development in certain areas of the animals and not others. The discs that give rise to antennae were especially sensitive to transformation by SO into eye tissue. The scientists then used light and confocal microscopy to determine the exact position of the newly formed retinal tissue in these discs.
Kumar says he and the members of his research group are hoping this avenue of experimentation will begin to provide answers to many questions, such as how a multicellular organism directs eye formation in correct regions of the body.
Source: Indiana University