Tidy motor protein folds away when the job is done
A discovery by University of Leeds researchers has revealed how a motor protein shuts itself down and becomes compact when it has no cargo to carry. It then goes in search of more cargo, perhaps carried by other passing proteins.
The discovery is significant, because the protein, called myosin 5, is part of a large family of motor proteins involved in a wide range of important bodily processes, including hearing, muscle contraction, digestion and neural transmission – the delivery of messages from the brain to different parts of the body.
“Some fatal seizures can be caused by a breakdown in myosin 5 activity,” says Dr Peter Knight of the Leeds research team. “By increasing our understanding of the process, we can start to address the problems caused when it doesn’t work. Of course there are still many questions that need answering, but we now have a model on which to base potential bio-nanotechnology molecular systems that could replicate the process to deliver molecules of our choosing – such as targeted drug therapies – to specific sites.”
The new discovery builds on the existing work of the Molecular Contractility Group of the University’s Astbury Centre for Structural Molecular Biology, who together with colleagues from the National Institutes of Health in the US, first showed how myosin 5 ‘walks’ to its destination.
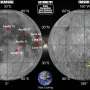
Myosin 5 molecules have two elongated heads attached to one end of a tail. Each head comprises a motor domain and a lever. At the other end of the tail is a pair of cargo-binding domains. In cells, myosin 5 is able to transport cargo because the two motor domains bind alternately to a filament called F-actin and take rapid strides along it by tilting the levers, like a pair of legs walking.
However, the filament it travels on is a one-way track, so myosin can only travel in one direction on its own. But once the cargo has been delivered – whether it is neurotransmitter chemicals to make a muscle contract or a package of pigments for a strand of hair – until now no-one knew what happened next.
Dr Kavitha Thirumurugan of the Leeds team commented: “It now seems that once its job is completed, myosin 5 folds up and the cargo binding domain does double duty by shutting down the motor, preventing myosin 5 from aimlessly wandering around. How it finds its next cargo we don’t yet know, but maybe it is picked up and carried there by proteins travelling in the other direction to start the cycle again. It’s a bit like a clockwork train network.”
Source: University of Leeds