Fundamental Discovery About the Fracture of Human Bone: It's All in the 'Glue'

A startling discovery about the properties of human bone has been made by scientists at the University of California, Santa Barbara.
The scientists describe their results –– finding a sort of "glue" in human bone –– in the cover story of the August issue of the international scientific journal, Nature Materials. The article was published on-line on July 17. It describes how healthy bone resists fracture and how unhealthy bone fractures at the molecular level. Included with the article are the highest resolution images of bone ever published, which reveal the location of the adhesive or "glue" that holds together mineralized collagen fibrils (protein fibers) of bone.
Image: Fracture surface of human bone showing mineralized collagen fibrils. Hansma Lab, UCSB
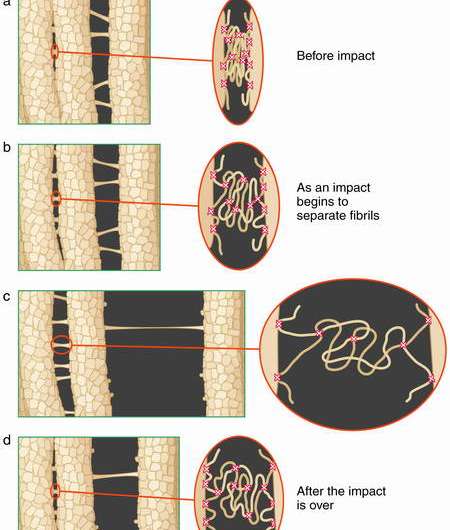
The glue appears to contain "springs" that uncoil when the bone is stressed, helping the bone to absorb shock. When the stress is relaxed, they coil back to their original structure.
The possible implications for human health are important, explained Georg E. Fanter, a UCSB doctoral student in physics and first author of the report. "The findings may lead to therapy for bone fracture, or even to prevention," he said.
Working in the physics laboratory of Professor Paul K. Hansma, in collaboration with the UCSB labs of Professors Daniel E. Morse and Galen D. Stucky, the interdisciplinary group of scientists spent several years tracking down where the glue was located in bone, and how it worked.
"Before this research, it was well known that the mechanical properties of bone depended on mineral particles and on collagen fibrils," said Hansma. "The picture of bone was that it consisted of these collagen fibrils coated with tiny mineral crystals only a few atoms thick. What we found is that there is a glue in bone that holds these mineralized collagen fibrils together, and this glue works along the same principles that our interdisciplinary research group found in abalone shells. This glue involves sacrificial bonds (with hidden length) that uncoil when the bone is stressed." That interdisciplinary research group included the research groups of Morse and Stucky, as well as that of Herbert J. Waite.
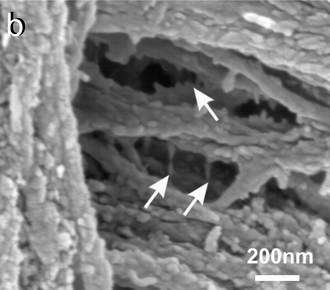
Image: Individual collagen fibrils are held together with glue filamets (arrows). Hansma Lab, UCSB
Said co-author Daniel Morse, director of UCSB's Institute for Collaborative Biotechnologies: "It's especially exciting for us to find the profound medical significance of our discoveries for human bone." He described the discovery of "molecular shock absorbers" providing a kind of self-healing glue holding biological mineralized structures together when studying the abalone shell six years ago. "It's truly remarkable to find the same fundamental mechanisms operating in bone," said Morse.
He noted that these mechanisms give young healthy bone its tremendous resiliency and resistance to fracture, and actually help heal small microcracks soon after they're formed. "We're especially interested in learning how these molecules change and become depleted with age as well as in certain diseases," said Morse. "A potential benefit from these discoveries is the prospect that we might now learn how to protect bone from these deleterious changes, and perhaps actually reverse some of the changes."
Hansma explained, "The thing that's exciting about this research is that we've identified a mechanically important component of bone." When the exact molecules are identified, these can then become therapeutic targets, for example, diet, or drug therapy. He said the group has made a fundamental discovery of something that's important for bone fracture resistance that was previously unknown. Now the degradation mechanisms and deficiencies can be studied. "If you don't know something is important then you can't do anything about it. This is a fundamental and new discovery in an old and well-studied field," Hansma added.
Credit: Hansma Lab, UCSB
He noted that bone fracture is a serious problem for elderly people, especially those with osteoporosis. Bone fracture is one of the leading factors in decreasing quality of life for the elderly. "Cancer and heart disease can kill you but bone fracture and arthritis make you miserable," he said. "Once a serious bone fracture occurs, then quality of life goes down. Less than one-third of elderly women who have a hip fracture return to previous function. More women die within a year of hip fracture than die after a heart attack. So it's a very serious problem and a lot of work has been done to develop good diagnostics. The leading diagnostic is bone mineral density."
Measuring bone mineral density is valuable because it reveals how much bone a person has left. The amount of an individual's bone mineral density peaks around age 30, then decreases for the rest of his or her life. By the time the person becomes elderly, very serious bone loss may have occurred.
"As if this weren't bad enough, it is also true that the materials properties of the bone decrease with age," said Hansma. "Not only is there less bone, but what exists is less strong. The cause of this is not well understood. Our research is aimed at understanding that."
Hansma noted that bone has been studied extensively since Galileo. "Galileo wrote a really nice paper about bone, describing why elephants need thicker bones than small animals." He said that a paper on bone is published every six minutes. And although bone is extensively studied, little is known about how it works at the molecular level. "Our paper is the beginning research on this."
Hansma and his colleagues at UCSB have pioneered the use of the Atomic Force Microscope (AFM) in looking at the nano-scale level of biological materials –– down to a billionth of a meter. Before the use of the AFM, it was impossible to see these tiny molecular structures.
In addition to Fanter, Hansma, and Morse, co-authors include Tue Hassenkam, Johannesh H.Kindt, Leonid Pechenik, and, Jacqueline A. Cutroni of the Hansma Lab; James C. Weaver of the Institute for Collaborative Biotechnologies at UCSB; Henrik Birkedal and Galen Stucky of the Department of Chemistry and Biochemistry; and Geraldo A. G. Cidade of the Biophysics Institute Carlos Chagas Filho at the Federal University of Rio de Janeiro, Brazil.
Source: University of California, Santa Barbara


















