New molecule involved in the body's processing of dietary fat identified
UCLA investigators have identified a new molecule that may help regulate the delivery of fats to cells for energy and storage. Published in the April issue of the journal Cell Metabolism, the finding could lead to a better understanding of how we utilize fats from the foods we eat.
"We thought that we had figured out how the body digests and uses fats, but we have identified a completely new player in the game," said the study's author Anne Beigneux, assistant investigator at the David Geffen School of Medicine at UCLA.
Digested fats travel to the small intestine, where they are packaged into chylomicrons, which are large, spherical particles filled with triglycerides.
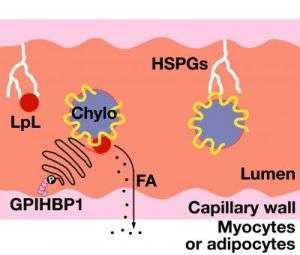
Chylomicrons then travel through the bloodstream and deliver triglycerides to the skeletal muscles and heart — tissues that are hungry for fuel — or to adipose tissue for energy storage. Molecules called proteoglycans, attached to the inside walls of capillaries, wait like baseball players with their mitts open, poised to catch the passing chylomicrons.
Proteoglycans hold the chylomicrons steady while the triglycerides are broken down or hydrolyzed by the enzyme lipoprotein lipase (LpL). The triglyceride breakdown products are then taken up and used by cells.
"Previously we didn't know what molecule in the capillaries facilitated the capture of chylomicrons and facilitated the interaction with lipoprotein lipase," said Dr. Stephen Young, study author and investigator at the David Geffen School of Medicine at UCLA. "We think that we've found the missing piece of the puzzle."
Investigators discovered that a protein called glycosylphosphatidylinositol-anchored high-density lipoprotein-binding protein 1 (GPIHBP1) may be the missing link.
Scientists found that mice deficient in GPIHBP1 develop very high triglyceride levels, even on a normal diet, demonstrating that fats in the bloodstream are not readily metabolized in the absence of GPIHBP1.
Laboratory tests confirmed that GPIHBP1-deficient mice had much higher levels of chylomicrons in the bloodstream than normal mice. The GPIHBP1-deficient mice had grossly milky plasma, reflecting very large amounts of triglycerides in the blood.
"These findings indicate a defect in the breakdown of chylomicrons in mice that don't have GPIHBP1," Beigneux said.
Investigators predicted that if GPIHBP1 were involved in the processing of chylomicrons in the bloodstream, then the protein would be made by endothelial cells of capillaries, where the breakdown of triglycerides takes place. Indeed, microscopy showed that GPIHBP1 is expressed highly and exclusively on the endothelial cells of capillaries of heart, adipose tissue and skeletal muscle.
Interestingly, scientists found that this protein was absent from the brain, which mainly uses glucose for energy.
"These differences suggest that endothelial cells may play an active role in regulating the delivery of lipid nutrients to different tissues," Beigneux said.
Experiments with cultured cells revealed that GPIHBP1 binds both chylomicrons and lipoprotein lipase, suggesting GPIHBP1 is a key platform for the processing of chylomicrons.
The next step, according to investigators, will be to determine if GPIHBP1 provides the only binding site for chylomicrons and lipoprotein lipase within capillaries. In addition, investigators would like to define the molecular basis for how GPIHBP1 binds to chylomicron particles.
Source: University of California - Los Angeles