Expression of a membrane protein in peripheral tissue linked to cancer: A novel tumour marker
Researchers at the Max Planck Institute of Experimental Medicine, in collaboration with the Department of Pathology at the Medical School of the Georg August University in Gottingen and the National Institute for Cancer in Rio de Janeiro, Brasil, have found a strong correlation of high expression of Eag1 potassium channels with multiple malignant tumour types. Their results have been published in two research articles published back-to-back in the 5th October 2006 issue of Molecular Cancer. This work on the innovative tumour marker, Eag1, opens the door to the development of novel strategies to diagnose and treat cancer and has high potential for clinical application.
In previous experiments, the researchers had shown that the Eag1 channel is frequently expressed in cell lines derived from tumours. They could also demonstrate that the ion channel is involved in cell-cycle progression in these cells, and that a significant reduction in cell proliferation could be achieved in vitro by inhibiting Eag1 expression with different methods. Additionally, the introduction of Eag1-transfected cells into mice caused rapidly growing and aggressive tumours, suggesting that Eag1 expression confers a selective advantage to cancer cells.
Ion channels are frequently implicated in a variety of pathological conditions, and they lend themselves as targets for therapeutic intervention because they are accessible molecules expressed on the surface of cells and relatively easy to target pharmacologically. A variety of ion channels have been reported to be associated with tumours, but Eag1 is the first case of an ion channel directly linked to tumour progression.
The use of reverse transcription real-time PCR and specifically generated and engineered monoclonal anti-Eag1 antibodies showed that the expression of the human Eag1 potassium channel (also known as KCNH1 or Kv10.1) is normally limited to specific areas of the brain and to very restricted cell populations throughout the body. This peculiar distribution adds to the interest of this particular channel as therapeutic target, because it could minimise undesired side effects.
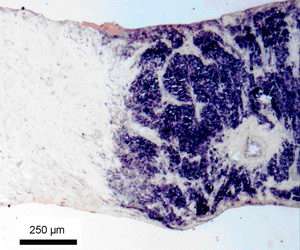
To evaluate the importance of Eag1 as a possible tumour target, the pattern and frequency of expression of the Eag1 channel in a large population of normal and tumour tissues from different collections was studied utilising molecular biological and immunohistochemical techniques. Pooled data from samples from 756 patients revealed intense overexpression of the channel in 77% of the cases. This is an extraordinarily high frequency, probably indicating that Eag1 expression confers better survival to tumour cells and that cells expressing the channel are selected for. Indeed, tumour samples taken several months after the first malignant changes have occurred are strongly enriched for Eag1-expressing cells.
In the accompanying paper, the same specific monoclonal antibodies were used to determine the expression levels of Eag1 in soft tissue sarcomas. Soft tissue sarcomas are rare tumours (less than 1% of all cancers) with bad prognoses (over 40% mortality in the first year after diagnosis). The relatively small number of cases and the great diversity in histopathologic presentation, anatomic site and biological behaviour has made a comprehensive understanding of these disease entities extremely difficult. Clinicians and patients are still faced with limited therapeutic options that deliver only a modest improvement in survival.
Eag1 was expressed in 71% out of 210 sarcomas studied. The frequency of Eag1 overexpression ranged from 56% (liposarcoma) to 82% (rhabdomyosarcoma). Differences in expression levels could be correlated only with the histological type, but not with either the sex or age of the patients, nor with the grade or tumour size. As for the previous study, the cell types that give rise to the sarcomas are always negative for Eag1 expression.
As examples for relevant sarcoma histological types (fibrosarcoma and rhabdomyosarcoma), four cell lines were tested as positive for Eag1 expression by real-time RT-PCR. Additionally, abolition of Eag1 expression in these cells by RNA interference led to a decrease in proliferation.
Taken together, these results indicate a direct involvement of Eag1 channels in cell proliferation and suggest that they participate in the transformation of normal cells into tumour cells. Outside of the brain, Eag1 channels are present only at very low levels in very restricted cell types. In contrast, tumours of many diverse types express Eag1 channels with greater frequency than other common tumour markers. Therefore, Eag1 represents a new target for cancer detection and diagnosis. In addition, it is a transmembrane protein and therefore accessible from outside the tumour cells.
Normal cells that express Eag1 are either protected by the blood-brain barrier or represent the terminal stage of normal differentiation. These facts together with the knowledge that Eag1 inhibition causes a reduction in tumour growth, means that this channel protein represents a promising target for the tailored treatment of human tumours and such therapies should produce only minor side effects.
Citation:
Hemmerlein, B., R.M. Weseloh, F. Mello de Queiroz, H. Knötgen, A. Sánchez, M. E. Rubio, S. Martin, T. Schliephacke, M. Jenke, H-J. Radzun, W. Stühmer, L.A. (2006) Pardo, Overexpression of Eag1 potassium channels in clinical tumours, Molecular Cancer 5:41
F. Mello de Queiroz, G. Suarez-Kurtz, W. Stühmer and L.A. Pardo (2006)
Ether à go-go potassium channel expression in soft tissue sarcoma patients, Molecular Cancer 5:42
Source: Max-Planck-Institute for Experimental Medicine