May 28, 2010 report
Arizona man is first to go home with a total artificial heart
(PhysOrg.com) -- An Arizona man has left hospital with a completely artificial heart beating in his chest. Father of three Charles Okeke, 43, from Phoenix is the first person to leave hospital with a Total Artificial Heart keeping him alive until he receives a donor heart.
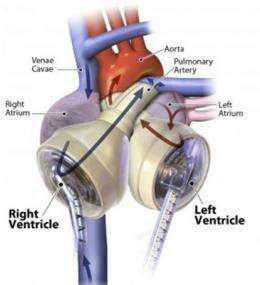
The US Food and Drug Administration (FDA) first approved the artificial heart for implantation into humans in 2004, and since then around 850 people around the globe have received them. The device uses synthetic pumping chambers to replace the failing left and right ventricles of the recipient’s own heart.
The artificial heart is designed to be used temporarily until a donated heart is available, and recipients have all had to remain in hospital while the heart was fitted, in some cases as long as two and a half years. The artificial heart is used only in patients who have both ventricles failing and enables 80 percent of them to survive long enough to receive the donated heart.
The artificial heart is made in Tucson, Arizona by SynCardia Systems. It consists of a vessel resembling an upside-down funnel and a flexible diaphragm. Surgeons replace each existing ventricle with a vessel and remove all four heart valves. They then hook up the artificial “ventricles” to the cardiovascular system, with the right vessel receiving blood and pumping it to the lungs for oxygenation, and the left pumping the oxygenated blood to the rest of the body.
The device requires pressurized air from an external machine called “Big Blue,” and the bulk and size of the 190 kg machine has until now prevented recipients from leaving hospital. In the case of Charles Okeke a smaller 6.1 kg version of the pressurizing machine, dubbed “Freedom Driver,” has been used, and this is small enough to fit in a backpack.
Okeke must wear the backpack at all times, but it has at least given him the freedom to leave hospital and go home, after already spending over 600 days in the Mayo Clinic wired up to Big Blue. Okeke’s heart failed after a blood clot “destroyed his heart” and had a heart transplant that lasted around 10 years before his body rejected it.
According to SynCardia, the company now has permission to use the Freedom Driver with 60 more patients. Of these, half will remain in hospital and half will be allowed to go home, which will allow the scientists to compare the health outcomes of the two groups.
SynCardia spokesman Don Isaacs said the artificial ventricles are designed to prevent blood clots forming, but with any device in which tubes pass from outside the body to the inside there is always a risk of infection.
More information: www.syncardia.com/Total-Artificial-Heart.html
© 2010 PhysOrg.com















