April 19, 2010 weblog
Could Printers Produce Human Tissue?
(PhysOrg.com) -- Scientists are getting closer to be able to create human tissue using special 3D printers. Using patients' own cells, the start-up company Organovo would use adipose tissue or bone marrow to craft new tissue. The adipose tissue would come from patients' own body fat stores, or bone marrow might be used instead. With these cells providing information specific to the patients' bodies, it would then be possible to create new tissue to replace the old.
The technique is known as bioprinting, and it makes use of special 3D printers, attached to computers running bio-programs, to help fabricate small sections of human tissue. In the future, it might even be used to fabricate new blood vessels or even nerves. CNET News offers this information on the possible applications of bioprinted human tissue:
As of right now, the benefit for humans is still years away, perhaps as many as four, said Organovo CEO Keith Murphy. And when and if the company's technology gets certified and hits the market, it will probably have limited application: most likely, the technology could be used at first mainly for crafting very small areas of tissue or new blood vessels.
But even those limited applications could mean, for example, that doctors may eventually have the ability to intervene in cases where, for example, a patient has a blocked or damaged blood vessel, and potentially prevent what might otherwise result in a forced amputation. Similarly, someone with damaged nerves could have a gap in a nerve bridged using regenerated cells printed by Organovo's machine.
Even farther into the future, there are hopes that such a technique could actually be used to create new organs, such as a replacement liver or a kidney. Of course, creating an entire organ in this manner will take time. The complexities involved in creating small swatches of human tissue in general are numerous. Creating an organ capable of full functionality would require even more complex processes and technology. One of the challenges is the necessity of being able to lay a vascular foundation that conducts life-giving blood to the tissue.
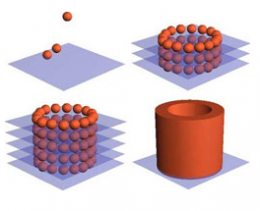
But, before the point of producing small tissue samples is even reached, one must figure out how to actually use a computer program and a bioprinter to build these small systems. CNET News describes the process by which bio-ink cells should create the required structure:
The key to the process is a fusion of the full cellular aggregate. Murphy explained that Organovo's 3D printers--which are built by the Australian firm Invetech, using Organovo's specifications--lay out a bio-ink made entirely of aggregates of cells, which within a period of about 24 hours of being prone, fuse together.
According to an Organovo white paper, "Cells attach to other cells, and cells produce collagen and attach to collagen. Cells know exactly how to behave once placed in the right orientation by the printer. They behave the same way they do in the body based on their inherent genetic programming."
Ultimately, the idea is that the printer pushes out fused cellular aggregates and lines them up "like balls in a paintball gun," Murphy said, and then deposits them very gently, according to a design that a doctor can define with a "very simple script."
Since the human tissue involved would be made with a patient's own cells as a blueprint, rejection of the new tissue would be less likely. One of the biggest problems with tissue transplant is rejection, in which the body attacks what it views as foreign. However, these bioprinted cells would most likely not be seen as foreign invaders, and could increase the rate of acceptance.
However, before any of this becomes a true reality, there are technological hurdles to overcome. But the idea is catching hold, and bioprinting may end up being helpful in the general search for better regenerative medicine.
More information: Daniel Terdiman, "Scientists edge closer to printing human tissue," CNET News (April 19, 2010). Available online: news.cnet.com/8301-13772_3-20002741-52.html
© 2010 PhysOrg.com














