'Relaxation' a critical step in vertebrate brain development
(PhysOrg.com) -- Normal vertebrate brain ventricle formation relies upon the stretchiness or "relaxation" of the neuroepithelium, which is regulated by the motor protein myosin. This process was discovered in zebrafish and may play a role in the proper expansion of tubes in other organs throughout development.
In their quest to unravel the mysteries of vertebrate brain development, Whitehead Institute researchers have discovered a novel mechanism integral to ensuring proper expansion and structural formation of the embryonic brain.
The researchers, who report their findings in the March 1 issue of Development, have dubbed their discovery "epithelial relaxation" — a lessening of rigidity in the cellular sheet that forms the future brain.
The vertebrate brain, including that of fish and humans, forms from a tube, the neural tube. During brain development, the center of the neural tube fills with embryonic cerebrospinal fluid (eCSF), to form a system of cavities known as brain ventricles. eCSF and brain ventricles are essential for normal nervous system formation and function. Scientists studying this process in zebrafish have observed that embryos lacking eCSF fail to fill their ventricles. However, curiously, some genetic mutant zebrafish strains produce eCSF, but fail to form ventricles. This suggests that a process in addition to eCSF production is required to expand the neural tube and form the brain ventricles.
“Almost every organ is a tube, whose cavity (lumen) must be correctly sized to carry normal amounts of fluid, so that the organ functions normally. In the brain, correct eCSF volume is essential for subsequent brain development and function," says Whitehead Member Hazel Sive. "For example, in hydrocephalus, too much eCSF results in severely impaired nerve cell formation and brain function. Our findings therefore expand understanding of why the brain may develop abnormally, and suggest new strategies by which other organs may develop.”
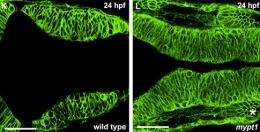
A major clue to this additional process was deduced by Jennifer Gutzman, a postdoctoral scientist in the lab of Whitehead Member Hazel Sive. Zebrafish with a mutation in the mypt1 gene, which codes for the Mypt1 protein (a critical regulator of the major motor protein myosin), fail to normally expand their neural tube, and form only small brain ventricles.
In order to rule out the possibility that insufficient eCSF causes the small ventricles in mypt1 mutants, Gutzman attempted to force the ventricles open with a high-pressure injection of fluid into the ventricular cavity. In the process she discovered that the neural tube in the mutant fish was not as "stretchy" as in normal fish, meaning that the ventricle space could not be forced to open fully. Gutzman particularly focused on the hindbrain ventricle, in the region of the brain that will give rise to the essential cerebellum and medulla of the adult. Gutzman and Sive make the analogy to blowing up a balloon.
Comments Sive: “A regular balloon expands not only because you are blowing air (or liquid) into it, but because the walls can stretch to accommodate the air (or liquid). You cannot blow up a PVC pipe, of course, because the walls of the pipe do not stretch. We thought that this analogy might extend directly to the brain tube, and that this needs to be stretchy in order to expand with eCSF.”
Having observed that the hindbrain ventricle walls — the neuroepithelia — in the mutant fish simply couldn’t be stretched as much as those in normal fish, Gutzman investigated whether the cells composing the neuroepithelia in the mutants might be different. Indeed, they were.
“The neuroepithelium is really complicated,” says Gutzman, who relied on sophisticated imaging and three-dimensional reconstruction techniques to observe brain individual cells in the living embryo. “We found that the cells in the mutants were short and squat, and rather stubby, compared with the cells in the normal fish, which are narrower, long, and spindly.”
The squatter shape of the cells offered a clue as to why the neuroepithelium might be less flexible. Though the specific mutation in question affects myosin phosphatase, myosin phosphatase ultimately regulates the activity of myosin. Myosin activation causes contraction, cellular tension, and rigidity. Therefore, Gutzman and Sive hypothesized that loss of myosin phosphatase would result in “hyperactive” myosin, and that in the mutants excessive myosin contraction may be responsible not only for abnormal cell shape, but also for inability of the ventricles to expand fully.
In order to test this theory, Gutzman exposed mutant embryos to blebbistatin, a drug known to inhibit the activity of myosin. After three hours of treatment with the drug, ventricles in the mutant fish were fully opened and normal shape was restored. Blebbistatin treatment also rescued the abnormal cell shapes in the mypt1 mutants, showing that myosin contractility has to be suppressed for both normal cell shapes and normal ventricle expansion.
Gutzman and Sive conclude that the neuroepithelium must relax, through reduction of myosin activity, for the hindbrain ventricle to reach its normal working volume. Epithelial relaxation is a critical step in early vertebrate brain development. It’s a finding that, according to Sive, has implications for understanding nervous system development and, potentially, the formation and function of other organ systems.
Says Sive: “Almost every organ is a tube, whose cavity (lumen) must be correctly sized to carry normal amounts of fluid, so that the organ functions normally. In the brain, correct eCSF volume is essential for subsequent brain development and function. For example, in hydrocephalus, too much eCSF results in severely impaired nerve cell formation and brain function. Our findings therefore expand understanding of why the brain may develop abnormally, and suggest new strategies by which other organs may develop.”
More information: “Epithelial relaxation mediated by the myosin phosphatase regulator Mypt1 is required for brain ventricle lumen expansion and hindbrain morphogenesis”, Development, March 1, 2010, 137; 795-804.
















