Study: 'Smart drug' Provigil may be habit-forming
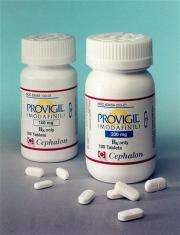
(AP) -- A so-called "smart drug" popular with young people may carry more of an addiction risk than thought, a small government study suggests. Scans of 10 healthy men showed that the prescription drug Provigil caused changes in the brain's pleasure center, very much like potentially habit-forming classic stimulants. Modafinil, the drug's generic name, is sometimes used as an illegal study aid by college students.
"It would be wonderful if one could take a drug and be smarter, faster or have more energy," said Dr. Nora Volkow, director of the National Institute on Drug Abuse, who led the study with a Brookhaven National Laboratory scientist. "But that is like fairy tales. We currently have nothing that has those benefits without side effects."
The study, appearing in Wednesday's Journal of the American Medical Association, may bust the myth that the drug is safe for healthy people, experts said.
Provigil is approved to treat excessive daytime sleepiness caused by narcolepsy. On the market since 1999, it's the flagship product of Cephalon Inc. of Frazer, Pa., and its sales approached $1 billion last year. The company is developing a spin-off called Nuvigil.
Modafinil's reputation as a brain enhancer stems from an Air Force study that found it improved the performance of sleep-deprived fighter pilots. College students buy and sell it illegally, as they do Ritalin and Adderall, to stay alert while studying.
Several scientists recently wrote in the journal Nature that healthy people should have the right to boost their brains with pills like Provigil. One author of that commentary, brain scientist Martha Farah of the University of Pennsylvania, said the new study "goes to show that we need a little caution and a little humility when we're messing around with our brain chemistry."
"But even now, after all the years that it has been on the market, we are still learning things about it that are relevant to its safety," Farah said.
The men in the study were 23 to 46 years old. They received either a dummy pill or modafinil. Effects were measured by PET scans, which showed that the drug increased dopamine, the brain's "feel-good" neurotransmitters.
Modafinil once was thought to be safer than conventional stimulants because it was believed that it did not engage the brain's dopamine system, which is linked with addiction. Studies in mice and monkeys suggested otherwise.
The new study is the first human evidence that a typical dose of modafinil affects dopamine in the brain as much as a dose of Ritalin, a controlled substance with clear potential for dependence.
Volkow said modafinil acts slowly when swallowed and is difficult to inject, making it less likely to be abused. Its high price, about $10 per pill compared to Ritalin at $2 per pill, also makes it less attractive to people seeking a high. That may change when generics become available in 2012, Volkow said.
Jeffry Vaught, chief science officer for Cephalon, said the company has seen no evidence the drug is highly abused.
"If abuse is a problem with modafinil, it's minimal at best," Vaught said. "We're not seeing it used at rave scenes."
Prescribing information for the drug warns of severe rashes and other side effects such as headache, nausea and anxiety. Cephalon doesn't support the drug's use as a cognitive enhancer.
"There's no substitute for sleep," Vaught said.
---
On the Net:
JAMA: http://jama.ama-assn.org
©2009 The Associated Press. All rights reserved. This material may not be published, broadcast, rewritten or redistributed.