Nasal Spray May End Dental Needle Injections for Upper Teeth Repair
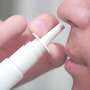
(PhysOrg.com) -- A nasal spray shown to numb the upper jaw is set to be tested in an FDA Phase 3 trial, which will assess the spray's effectiveness compared to the current "gold standard" treatment -- painful anesthesia injections.
"A successful trial of this new dental anesthetic will change dental technology worldwide," said Sebastian Ciancio, D.D.S., University at Buffalo SUNY Distinguished Service Professor and chair of the Department of Periodontics and Endodontics.
Ciancio directed the Phase 2 trial and will coordinate the Phase 3 trial. Between 6 and10 million dental needle injections are given daily, according to Ciancio.
Results of the FDA Phase 2 trial, conducted in 48 subjects at the UB School of Dental Medicine, showed that the spray appears to be safe and effective.
The Phase 3 trial will be carried out later in 2009 at the UB dental school and other clinical sites. Ciancio said that if the Phase 3 trial is successful, it may mean the end of injections for any dental work performed on the upper teeth.
Ciancio and colleagues conducted the initial preliminary dental studies using the nasal spray, which is being developed by St. Renatus, LLC, based in Fort Collins, Colo.
The nasal spray formula being tested is related to a drug used by ear, nose and throat physicians when they operate on the nose. Patients who received this anesthetic reported that their upper teeth felt numb, sparking interest in using the anesthetic for dental procedures. The spray is effective only on the upper teeth.
Provided by University at Buffalo