Scientists Learn How Some Cancer Cells Survive Chemo
What separates the few cancer cells that survive chemotherapy - leaving the door open to recurrence - from those that don’t? Weizmann Institute scientists developed an original method for imaging and analyzing many thousands of living cells to reveal exactly how a chemotherapy drug affects each one.
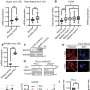
For research student Ariel Cohen, together with Naama Geva-Zatorsky and Eran Eden in the lab of Prof. Uri Alon of the Institute’s Molecular Cell Biology Department, the question posed an interesting challenge. To approach it, they needed a method that would allow them to cast a wide net on the one hand - to sift through the numerous cellular proteins that could conceivably affect survival - but that would let them zoom in on the activities of individual cells in detail, on the other.
Letting the computer take over the painstaking work of searching for anomalies enabled the team to look at the behavior of over 1,000 different proteins. Even so, it took several years to complete the project, which entailed tagging the specific proteins in each group of cancer cells with a fluorescent gene and capturing a series of time-lapse images over 72 hours. A second, fainter fluorescent marker was added to outline the cells so the computer could identify them. A chemotherapy drug was introduced 24 hours into this period, after which the cells began the process of either dying or defending themselves against the drug.
The team’s efforts have produced a comprehensive library of tagged cells, images, and data on cancer-cell proteins - a virtual goldmine of ready material for further cancer research. And they succeeded in pinpointing two proteins that seem to play a role in cancer cell survival.
Although most of the proteins behaved similarly in all the cells, the researchers found that a small subset of them - around five percent - could act unpredictably, even when the cells and drug exposure were identical. The scientists called these proteins bimodal, as they acted in one of two ways.
The team then asked whether any of the bimodal proteins they identified were those that occasionally promote cell survival. They found two molecules that seemed to fit the bill. One of them, known by the letters DDX5, is a multitasking protein that, among other things, plays a role in initiating the production of other proteins. The other, RFC1, also plays varied roles, including directing the repair of damaged DNA. When the researchers blocked the production of these proteins in the cancer cells, the drug became much more efficient at wiping out the growth.
Cohen: “This method gave us tremendous insight into how a cell responds to a drug. By conducting an unbiased study - we started with no preconceived notions of which proteins were involved - we were able to pinpoint possible new drug targets and to see how certain activities might boost the effectiveness of current drugs.”
Prof. Uri Alon’s research is supported by the Kahn Family Foundation for Humanitarian Support and Keren Isra - Pa’amei Tikva.
The Weizmann Institute of Science in Rehovot, Israel, is one of the world’s top-ranking multidisciplinary research institutions. Noted for its wide-ranging exploration of the natural and exact sciences, the Institute is home to 2,600 scientists, students, technicians, and supporting staff. Institute research efforts include the search for new ways of fighting disease and hunger, examining leading questions in mathematics and computer science, probing the physics of matter and the universe, creating novel materials, and developing new strategies for protecting the environment.
Source: Weizmann Institute of Science