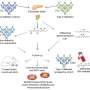
Computers make sense of experiments on human disease
Increased use of computers to create predictive models of human disease is likely following a workshop organised by the European Science Foundation (ESF), which urged for a collaborative effort between specialists in the field. Human disease research produces an enormous amount of data from different sources such as animal models, high throughput genetic screening of human tissue, and in vitro laboratory experiments. This data operates at different levels and scales including genes, molecules, cells, tissues and whole organs, embodying a huge amount of potentially valuable insight that current computer modelling approaches often fail to exploit properly.
However, significant advances in the modelling of a few specific diseases, such as multiple sclerosis (MS), have been made. A major aim of the ESF workshop was thus to generalise such work and create a more coherent body of expertise across the whole field of computational disease analysis, according to Albert Compte, co-convenor of the ESF workshop, from the Computational and physiological bases of cortical networks laboratory at the Institut d'Investigacions Biomèdiques August Pi Sunyer (IDIBAPS) in Barcelona. "A workshop like this one was useful in seeing how advances in other research fields can be used more generally for disease modelling," said Compte. "So far, novel modelling approaches have been confined to a specific disease or a particular level of description".
A model might be confined just to the molecular level or the cellular level for example. The ESF workshop highlighted the benefits that could be obtained from integrating data from different levels. This can provide more detailed and flexible models, with greater power to identify causes of diseases and predict possible cures in future.
However, one potential problem when building sophisticated disease models operating at different levels is that they can become too complex, with a lack of sufficient data for any useful analysis. This can be resolved by selecting a simpler model that corresponds only to the experimental data that actually exists. Delegates at the workshop heard how in the case of MS, selection of the model could be tuned to the data, to make best use of the actual experimental results obtained in a particular study, as explained by Jesper Tegner, another co-convenor of the ESF workshop, from the Atherosclerosis Research Unit at the Karolinska Institute Centre for Molecular Medicine (CMM) in Stockholm, Sweden.
"There was one exciting presentation on MS," said Tegner. "The immune system is clearly central for MS. However, the trick in the case of MS is to represent different aspects of the immune system according to the available data.The objective isn't to model the whole immune system. One interesting level of abstraction was the presentation of agent-based modelling of MS where individual cells operated as agents, thus omitting the intracellular machinery." In other words, the detailed interior workings of the cells could be ignored in this case because that would have made the model overcomplicated, with insufficient data at the different levels to produce any useful insights.
In other experiments, data about varying levels of gene expression was obtained, which required very different models with networks of graphs. These highlighted the patterns of gene expression associated with a particular disease, such as MS.
Yet another valuable application of computer-based mathematical disease models lies in studying the phenomenon of addiction to drugs such as nicotine and helping to reconcile conflicting theories, as Compte pointed out. "The neurobiology of nicotine addiction is a hotly debated field. In particular, there are two contending views on how neurons and their connections in subcortical nuclei are affected by nicotine. This computer model allows us to reconcile the apparently contradictory results obtained from in vitro and in vivo experiments, and thus provides a single theoretical proposal of how nicotine affects neuronal circuits in the brain and causes addiction, compatible with most available experimental results."
Tegner and others at the workshop were confident that a coherent framework for building multi-level mathematical models on the basis of available data will lead to better understanding of many diseases and conditions such as drug addiction. This in turn, could lead to better therapies.
Source: European Science Foundation