Researchers Find 'Junk DNA' May Have Triggered Key Evolutionary Changes in Human Thumb and Foot

(PhysOrg.com) -- Out of the 3 billion genetic letters that spell out the human genome, Yale scientists have found a handful that may have contributed to the evolutionary changes in human limbs that enabled us to manipulate tools and walk upright.
Results from a comparative analysis of the human, chimpanzee, rhesus macaque and other genomes reported in the journal Science suggest our evolution may have been driven not only by sequence changes in genes, but by changes in areas of the genome once thought of as "junk DNA."
Those changes activated genes in primordial thumb and big toe in a developing mouse embryo, the researchers found.
"Our study identifies a potential genetic contributor to fundamental morphological differences between humans and apes," said James Noonan, Assistant Professor of Genetics in the Yale University School of Medicine and the senior author of the study.
Researchers have long suspected changes in gene expression contributed to human evolution, but this had been difficult to study until recently because most of the sequences that control genes had not been identified. In the last several years, scientists have discovered that non-coding regions of the genome, far from being junk, contain thousands of regulatory elements that act as genetic "switches" to turn genes on or off.
An indication of their biological importance, many of these non-coding sequences have remained similar, or "conserved," even across distantly related vertebrate species such as chickens and humans. Recent functional studies suggest some of these "conserved non-coding sequences" control the genes that direct human development.
In collaboration with scientists at Lawrence Berkeley National Laboratory in California, the Genome Institute of Singapore, and the Medical Research Council in the United Kingdom, Noonan searched the vast non-coding regions of the human genome to identify gene regulatory sequences whose function may have changed during the evolution of humans from our ape-like ancestors.
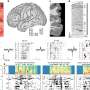
Noonan and his colleagues looked for sequences with more base pairs in humans than in other primates. The most rapidly evolving sequence they identified, termed HACNS1, is highly conserved among vertebrate species but has accumulated variations in 16 base pairs since the divergence of humans and chimpanzees some 6 million years ago. This was especially surprising, as the human and chimpanzee genomes are extremely similar overall, Noonan said.
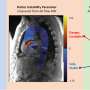
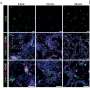
Using mouse embryos, Noonan and his collaborators examined how HACNS1 and its related sequences in chimpanzee and rhesus monkey regulated gene expression during development. The human sequence activated genes in the developing mouse limbs, in contrast to the chimpanzee and rhesus sequences. Most intriguing for human evolution, the human sequence drove expression at the base of the primordial thumb in the forelimb and the great toe in the hind limb. The results provided tantalizing, but researchers say preliminary, evidence that the functional changes in HACNS1 may have contributed to adaptations in the human ankle, foot, thumb and wrist-- critical advantages that underlie the evolutionary success of our species.
However, Noonan stressed that it is still unknown whether HACNS1 causes changes in gene expression in human limb development or whether HACNS1 would create human-like limb development if introduced directly into the genome of a mouse.
"The long-term goal is to find many sequences like this and use the mouse to model their effects on the evolution of human development," Noonan said.
Provided by Yale University