Study provides clues to preventing and treating cancer spread
Isn't it odd that cancer cells from one organ, such as the skin, can travel and take root in a totally different organ, like the lung?
What's more, why is it that certain cancers prefer to spread, or metastasize, to certain places? Prostate cancer usually moves to bone; colon cancer, to the liver.
To answer these questions, Dr. Hendrik van Deventer, assistant professor of medicine at the University of North Carolina at Chapel Hill and a member of the UNC Lineberger Comprehensive Cancer Center, turned to a century-old idea of cancer spread: English surgeon Stephen Paget's "seed and soil."
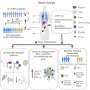
The idea is that the spread of cancer isn't just about the tumor itself (the seed), but also the environment where it grows (the soil). Other scientists have shown that cells from bone marrow can migrate and change the environment so that it is receptive to incoming cancer cells. These cells do so by forming small neighborhoods or niches within distant organs. Thus, biologists refer to these areas as "premetastic niches."
Van Deventer and his colleagues wanted to know what mysterious non-tumor cell could change a normal organ so cancer cells would invade. If scientists could discover the identity of that normal cell, maybe they could devise treatments to stop metastases.
In a study published in the July issue of The American Journal of Pathology, van Deventer showed for the first time that that cell could be a fibrocyte – cells that travel around the body, rushing to the site of an injury to aid in healing when needed. The study also suggests ways to develop treatments to prevent metastases using already available medications.
"This study shows it's possible for fibrocytes to form the premetastatic niche. But it stops short of proving they positively are the cells," van Deventer said.
The UNC researcher's work with fibrocytes began when he wanted to figure out why "knockout mice" that are missing the cell receptor CCR5 get fewer cancer metastases than normal mice. CCR5 helps control the migration of cells through the body. He injected these knockout mice with all types of cells from normal mice, to try to make the mice form more metastases of melanoma (skin cancer).
The only cells that did it were those that appeared to be fibrocytes.
When van Deventer injected the mice with just 60,000 of these cells, the rate of metastases nearly doubled. "That's a big effect for a relatively small number of cells," he said.
Though cancer researchers don't usually study fibrocytes, it makes sense to van Deventer that fibrocytes could form the premetastatic niche. In healthy humans, fibrocytes travel through the bloodstream to areas of injury. Once there, they produce changes that are good for wounds. Unfortunately, these same changes can help cancers grow. It is not yet clear if fibrocytes are causing these problems in cancer patients. However, "there is some clinical data that suggests that these cells are increased in patients with metastatic cancer," he said.
The experiment also showed that injection of these cells induced MMP9, an enzyme that is known to promote cancer. The researchers considered this good news, since drugs are available that block MMP enzymes and have proven beneficial in treating cancer.
Still, many basic questions remain to be answered. How do cancers promote the formation of the premetastatic niche? Do they change the behavior of these circulating cells or simply increase their number? Are some patients at higher risk for metastasis because their environment changes their fibrocytes? Is some of the benefit of our cancer treatments lost because of inadvertent changes to these cells?
"These are daunting questions, but ones that would have pleased Dr. Paget," van Deventer said. "This paper gives us a place to start looking for the answers."
Source: University of North Carolina