Key step in the 'puncture' mechanism of cell death revealed
A team of medical researchers led by Dr Ruth Kluck at Melbourne’s Walter and Eliza Hall Institute (WEHI) has discovered a key step in the mechanism by which cells destroy themselves. In this process, called “apoptosis”, certain proteins cause the cell to self-destruct by puncturing its “power plant.” How the proteins do this has been clarified by the WEHI team. The discovery is an important step towards the identification of targets for drugs designed to regulate cell death.
Dr Kluck and her colleagues explore how cells engineer their own destruction. Properly regulated cell death is actually essential for good health. This is because our cells naturally have a limited life span. The worn out, damaged or unnecessary cells in our bodies are eliminated at the rate of one million per second and replaced by the same number of new cells for as long as we live.
If a cell fails to die when it reaches its “use-by” date, it may go on to multiply uncontrollably and form a cancer. Conversely, a person in whom too many cells self-destruct may develop a degenerative disease such as Alzheimer’s.
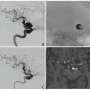
The cell’s self-destruction is driven by a protein called Bak that acts by puncturing the membrane of the mitochondria - the cell’s power plant. Once this power plant is wrecked, the cell is doomed.
But how does Bak do this? The crux seems to be the way Bak can bind to itself and form complexes that damage the mitochondrial membrane. Dr Kluck and colleagues have discovered that cellular distress signals cause one segment of Bak to flip out and insert neatly into a groove on another Bak molecule. The Bak doublet then forms the larger complexes that can puncture the mitochondria and provoke the cell’s self-destruction.
This insight into how apoptosis starts will assist in the development of drugs that can flick on the apoptosis “switch” to kill cancer cells more effectively. The development of drugs with the opposite effect is also important: to “switch off” unwarranted apoptosis in degenerative disorders.
Source: Research Australia