Study suggests new treatments for Huntington's disease
Working with fruit flies, researchers have discovered a new mechanism by which the abnormal protein in Huntington’s disease causes neurodegeneration. They have also manipulated the flies to successfully suppress that neurodegeneration, which they said suggests potential treatments to delay the onset and progression of the disease in humans.
Juan Botas and colleagues published their findings in the January 10, 2008, issue of the journal Neuron, published by Cell Press.
Huntington’s disease is caused by a mutation in the gene for the huntingtin protein (htt) that causes a genetic “stutter”—an abnormally long number of repeats of the amino acid glutamine at one end of the protein.
Previous studies had concentrated on the toxicity that the abnormal protein produces by forming cell-clogging aggregates in the nuclei of neurons. Most studies in animals, however, had not involved introducing the gene for full-length htt; they involved only a fragment.
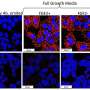
In their experiments, Botas and colleagues introduced the gene for full-length abnormal human htt into the fruit fly Drosophila and studied its early effects on neural function in the flies.
They found that, before the abnormal protein produced any toxic effects in the nuclei of neurons, it caused abnormally high transmission of chemical signals, called neurotransmitters, among neurons. Such neurotransmitters are launched by one neuron across connections, called synapses, to its neighbor, triggering a nerve impulse in the receiving neuron. Besides abnormal synaptic transmission, the researchers also found that mutant htt caused neurodegeneration and degeneration in the flies’ motor ability.
The researchers found that they could suppress these abnormalities by introducing other mutations into the fly genome that either reduced neurotransmission or reduced the activity of pores called calcium channels in the membranes of neurons. Such channels trigger neurotransmission by controlling the influx of calcium into neurons.
“The findings described in this report unveil a mechanism of pathogenesis for expanded htt that does not require its nuclear accumulation in detectable amounts,” concluded the researchers. They wrote that the increased neurotransmission they detected “likely represents a mechanism of pathogenesis taking place at early stages of disease progression.
“These findings point to increased synaptic transmission as a therapeutic target with the potential of delaying [Huntington’s disease] onset and thus likely impacting disease progression,” they wrote. They concluded that their ability to genetically suppress the abnormal neurotransmission and neurodegeneration “further define[s] specific therapeutic targets and support[s] the idea that Ca2+ channel antagonists, and perhaps other inhibitors of neurotransmission, offer an attractive therapeutic option due to their specificity and wide usage.”
Source: Cell Press