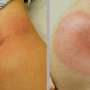
Freeze-dried tendon implants prove effective in early studies
Donated, freeze-dried tendon grafts loaded with gene therapy may soon offer effective repair of injured tendons, a goal that has eluded surgeons to date. According to study data published today in the journal Molecular Therapy, a new graft technique may provide the first effective framework around which flexor tendon tissue can reorganize as it heals.
Such tissue-engineering approaches could significantly improve repair of anterior cruciate ligaments and rotator cuffs as well, researchers said. The study was in a mouse model designed to resemble hard-to-repair flexor tendons in human hands, and the results should provide an impetus for future clinical trials.
Tendons are elastic cords that anchor muscle to bone and enable flexing muscle to move limbs. Related injuries represent nearly half of 33 million U.S. orthopaedic injuries each year, and a frequent cause of emergency room visits. In many standard repair attempts, surgeons implant an autograft, a piece of tendon from elsewhere in the same patient. Along with requiring patients to sacrifice tendon, the problem with “live” autografts is that both the graft and the graft site “know” they have been injured. That signals immune cells and chemicals to rush into the graft site, seeking to fight infection. Unfortunately, those same processes cause inflammation and scarring, which in turn cause implanted tendon to stick to the joint. To work properly, the tendon must be free to glide across the joint. Tendon adhesions, a longstanding post-surgical problem, cause pain and permanently limit range of motion.
Researchers next experimented with allografts: tendons donated from one person to another. Clinically, this technique fared worse than autografts because patients’ bodies would recognize the donated tendon as foreign, attempt to wall it off with fibrous proteins and in some cases reject the transplant. The field then looked at whether synthetic scaffolds made of gel or fiber mesh could serve as alternatives. Theoretically, such materials would guide damaged tissue as it reorganizes into healthy tendon without causing an immune reaction. They could be coated with anti-inflammatory drugs, growth factors or gene therapy vectors to drive healing and reduce swelling. Unfortunately, artificial grafts too failed to yield useful tendon substitutes because they did not match the mechanical strength of human tissue.
In the newly published study, a research team from the University of Rochester Medical Center explored yet another option: the implantation of allografts (donated, freeze-dried tendon) loaded with gene therapy. Their results show that the allografts served as effective tissue-engineered scaffolds, with significantly fewer adhesions than seen with autografts. The allografts also sucked up, and delivered into the graft site, a solution of gene therapy vectors that directed the recipient’s cells to accept the graft and remodel it into living tissue.
“Orthopaedic surgeons have been searching for the perfect material to replace tendon, one with the right mix of strength and elasticity and would not cause adhesion,” said Hani Awad, Ph.D., assistant professor of Biomedical Engineering and Orthopaedics within the Center for Musculoskeletal Research at the Medical Center. “We believe the only material to meet these strict requirements is non-living, but structurally intact tendon. We were surprised to find that no one had tried combining it with gene therapy or other drug delivery techniques to overcome its limitations,” said Awad, also senior author of the study.
Study Details
Tendon, like bone and cartilage, is connective tissue made up of tough protein fibers. The quality that enables tendon allografts to overcome past limitations is that such connective tissues naturally contain depots designed to hold signaling molecules. In the current study, tissue engineers filled those depots with gene delivery vectors.
In general, gene therapy inserts genes into cells, where they direct the target cell’s own genetic machinery to make a desired protein. In the current study, the inserted gene called for the building of a growth factor that directs cells to divide and tissues to grow, or heal. To deliver genes into cells, gene therapies rely on viruses (vectors) designed by evolution to penetrate human cells and insert their own DNA. Viral vectors retain this ability, but have been harnessed to deliver therapeutic genes. Specifically, Awad’s team implanted into the distal flexor digitorum longus (FDL) tendons of mice a freeze-dried allograft loaded with a recombinant adeno-associated vector (rAAV) expressing the gene that codes for the building of growth and differentiation factor 5 (Gdf5). A control group received an allograft loaded with a non-therapeutic gene (lacZ). Functional recovery was then compared between groups.
In past studies, rAAV vectors have proven to be safe because they make temporary changes to DNA, but then stop before too much re-growth can pose cancer risk. GDF5 was chosen because it is known to direct the formation of tendon in the womb. Similar to skin, tendons heal via the formation of a scar, but that process in tendon leads to imperfect tissue growth that adheres to the joint and compromises function. The hope was that adding extra GDF5 would help, and the data indeed show that animals with freeze-dried FDL allografts loaded with rAAV Gdf5 recovered twice the range of motion when compared to the control group at 14 days post surgery. At 28 days after surgery, the allograft group had reached nearly 65 percent of the normal range of motion, compared to the control group, which had recovered only 35 percent of the normal range.
Current rehabilitation programs take advantage of the fact that the gliding and stretching of tendon as it heals has been shown to accelerate healing. Various forms of passive, controlled motion (physical therapy) are commonplace. A limitation of the current study was that the mouse tendon allografts used were so small that the tendon had to be immobilized during the healing process to prevent tearing. Thus, the results showed that overall healing of the two groups – GDF-treated and control – proceeded at the same rate over the first 84 days after reconstruction. In larger animals and in humans, where allografts should be able to benefit from the force of motion as they heal, Awad expects that gene-therapy-loaded allografts will heal at a much faster rate than autografts or synthetic grafts. That theory has yet to be proven however.
Should this line of work prove successful, existing tissue banks could be refitted to create a nationwide supply of therapeutically enhanced tendons for transplant, according to the study authors. Millions of bone and cartilage grafts are already used in orthopaedics, as well as in plastic and general surgery. The banks are made possible by conscientious donors that indicate in their wills, or on their licenses, that their tissue is to be donated upon their death.
Along with Awad, study authors were Patrick Basile, M.D., Tulin Dadali, B.S., Justin Jacobson, M.D., Yasuhiko Nishio, Ph.D., M. Hicham Drissi, Ph.D., Howard Langstein, M.D., David Mitten, M.D., Regis J O’Keefe, M.D., Ph.D., and Edward Schwarz, Ph.D. from the University of Rochester Medical Center as well as Sys Hasslund, Michael Ulrich-Vinther and Kjeld Søballe from Aarhus University Hospital in Denmark. The team will next seek to determine the mechanisms by which growth factors repair tendons. After that, studies will move into larger animals and humans, potentially within a few years.
Source: University of Rochester Medical Center