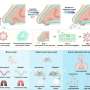
Copy number variation may stem from replication misstep
Genome rearrangements, resulting in variations in the numbers of copies of genes, occur when the cellular process that copies DNA during cell division stalls and then switches to a different genetic “template,” said researchers at Baylor College of Medicine in Houston in a report that appears today in the journal Cell.
The new mechanism is called replication “Fork Stalling and Template Switching,” said Dr. James R. Lupski, Cullen professor of molecular and human genetics and vice chair of the department at BCM. He is also professor of pediatrics. It not only represents a new way in which the genome generates DNA copy number variation, but it also demonstrates that copy number variation can occur at a different time point in the life of a cell. DNA replication takes place as the cell is dividing and becoming two.
Copy number variation involves structural changes in the human genome that result in the deletion or extra copies of genes (or parts of them). Often, this process is associated with disease, and also with evolution of the genome itself.
DNA (deoxyribonucleic acid) exists as two complementary strands that remain together because of the attraction between nucleotides. A or adenosine is always attracted to T (thymidine). C or cytosine is always attracted to G or guanine.
When a cell divides, it must reproduce its DNA so that each cell that results from the division has the same genetic code. That means it must replicate its DNA. During this process, an enzyme called a helicase separates the two strands, breaking the hydrogen bonds between the A – T and G – C base pairs holding the strands together. The two separating strands become the replication fork. On one strand, an enzyme called DNA polymerase reads the genetic material in the strand as a template and makes a strand (leading strand) of complementary DNA to pair to it. Again, the code is A to T and C to G. This process is continuous. On the other strand that comprises the fork, the complementary strand (lagging strand) is made in short, separated segments by a process that involves RNA and a series of enzymes.
Until the 1990s, researchers studying reasons for genetic mutations or changes looked at molecular “typos” in this process, tiny changes in the As, Ts, Cs or Gs called single nucleotide polymorphisms (SNPs). These SNPs changed the message of the gene. However, in the early 1990s, Lupski was one of the pioneers to elucidate a new mechanism in which the structure of the DNA itself was grossly duplicated or deleted, which changed numbers of copies of a gene that occurred in this genetic material. This “copy number variation” wrote a new chapter in the understanding of genetic variation.
Lupski and his former graduate student Dr. Jennifer Lee (now a postdoctoral fellow at BCM) found in their experiments that this process stalls when there is a problem with the DNA. In that case, the process switches to a different template, copying another similar but significantly different stretch of DNA, before it switches back to the appropriate area. Dr. Claudia M. B. Carvalho, also of BCM, took part in this research.
Previously, Lupski and colleagues had identified two different ways in which recombinations of genetic material resulted in copy number variation. However, when Lee was studying an inherited disease called Pelizaeus-Merzbacher disease, she found changes in the genome that the previous theories about DNA recombination did not explain.
Structural changes in the genome in people with the disease, a neurodevelopmental disorder, vary from person to person. In some places, genetic material that was duplicated was similar to that nearby but it was thrust into the middle of another duplication of material. The question was how they got there, Lee said.
The fork stalling, template switching mechanism explained the oddities, said Lupski.
“It stalls and rather than restart at the position where it is, it switches to a different template,” said Lupski. Usually this occurs in an area of the genome where there are many repeats of the nucleotides that form an unusual structure. This can actually aid in the template switching, he said.
“One could envision that it could happen anywhere in the genome and would be a way to make copy number changes in any gene you want,” he said. It might even play a role in evolution, allowing organisms to change. Some of the changes might make it easier for that organism to live in particular environment or survive in a stressed situation.
Source: Baylor College of Medicine