Life-threatening gene defect located by UT-Houston researchers
A research team led by scientists at The University of Texas Medical School at Houston has identified a defective gene that affects vascular smooth-muscle cells in people who suffer from hereditary thoracic aortic disease, which can kill victims with little warning in the prime of their lives.
Thoracic aortic disease, specifically thoracic aortic aneurysms leading to aortic dissections, is the 15th leading cause of death in the country, killing up to 20,000 people a year. Actor John Ritter (age 54 years) and “Rent” creator Jonathan Larson (age 35 years) both died from the disease. Cardiac surgeons in the Texas Medical Center – including Michael DeBakey. M.D., and Denton Cooley, M.D. – pioneered the surgical repair of thoracic aortic disease.
The study, “Mutations in Smooth Muscle Alpha-Actin (ACTA2) Lead to Thoracic Aortic Aneurysms and Dissections,” will be published in the Nov. 11 issue of Nature Genetics.
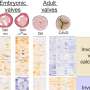
Research on 14 families with 93 members from across the United States, along with Europe and Australia, led the team to a mutation in the smooth muscle cell alpha-actin (ACTA2), which affects muscular contractions in other blood vessels and the aorta. The wall of the aorta, the main blood vessel leading out of the heart, weakens and then enlarges, forming an aneurysm and ultimately leading to an aortic dissection, which often causes sudden death.
“Our results suggest that contraction of the smooth muscle cells in the aorta is important in keeping it healthy and preventing the disease,” said the study’s senior author, Dianna M. Milewicz, M.D., Ph.D., professor and director of the Division of Medical Genetics at the UT Medical School. “This information provides insight into the cause of this condition and helps us understand, for the first time, the pathology of the aortic wall degeneration.”
Milewicz said that mutations in ACTA2 account for 14 percent of the inherited form of thoracic aortic aneurysms and dissections, making it the major gene identified for this condition.
“Dr. Milewicz has been an international leader in discovering gene defects that can cause vascular abnormalities,” said study co-author Dong Kim, M.D., chairman of the medical school’s Department of Neurosurgery and co-director of the Mischer Neuroscience Institute at Memorial Hermann. “This recent finding has enormous implications for diseases affecting blood vessels, from the aorta to the arteries in the brain. I believe that Dr. Milewicz’s work is going to lead to cures in the near future, and I feel privileged to be able to work with her.”
The aorta supplies blood to the rest of the body. Some people develop a progressive degeneration of the aortic wall, leading to a bulging aneurysm, which then can dissect, or tear.
Family members who test positive for the defective gene are encouraged to have routine imaging of their thoracic aortas in order to surgically repair an aneurysm before it dissects or ruptures. If caught early enough, when an aneurysm is 5 centimeters or less, a surgical procedure to replace the diseased portion with a Dacron graft has a high degree of success.
Early in her decade-long research research into the disease, which includes the discovery of two other genes linked to thoracic aortic aneurysms, Milewicz and her team took blood samples from a large family. For years they could not link them to any known gene defects until now.
Milewicz – who holds the President George Bush Chair in Cardiovascular Medicine – noted that members of this family with a known history of the disease had “livedo reticularis,” a purplish skin discoloration clearly visible on their arms and legs. Milewicz said that in families with a genetic background of the disease, livedo reticularis is a clinical sign of the disease that can alert physicians that the patient is at a higher risk for thoracic aortic aneurysms and dissections.
Families who carry the new gene have lost family members at tragically young ages. Houstonian Bruce Bryant lost his nephew Ian Bryant at age 17 in 2003. Bruce Bryant’s father died of heart disease at age 76, but no one linked those two deaths together.
Bryant, 51, began to feel pain in the center of his chest nearly two years ago while on a business trip to Oklahoma. During the flight home, he vomited and passed out. He was taken by ambulance to Memorial Hermann Northeast, where emergency room doctors quickly and astutely diagnosed his dissecting thoracic aorta.
He was sent by LifeFlight to Memorial Hermann – Texas Medical Center and delivered into the hands of surgeon Hazim J. Safi, M.D., professor and chairman of the Department of Cardiothoracic Vascular Surgery at the UT Medical School.
Blood from the dissected aorta began to fill up the sac around Bryant’s heart and his heart stopped beating. With no time for anesthesia, Safi, who also is chief of cardiovascular and thoracic surgery at Memorial Hermann, cut open his chest and punctured the sac to allow the blood to drain. Bryant’s heart began beating again but he had to wait a week until he was strong enough to have his dissection repaired.
When UT-Houston researchers approached Bryant about joining the study after he recovered, he didn’t hesitate. Family members in other states also agreed to participate. They learned that Bryant’s late brother, Robert (who died after an accident), carried the gene, as did his son Ian, who died very young of aortic dissection. Bryant has two sisters, both of whom carry the gene and one of whom has children who are being tested for carrying the defective gene. Bryant has no children.
“What shocked me about joining the study is that I thought I would hear something in 15 years. I didn’t expect to get a call in a year telling me that they had found the gene,” Bryant said. “The plan going forward is that the whole family has to get screened once a year. Even though it’s not a happy thing knowing that we carry it, everyone feels better that they know about it and can watch for it now.”
Source: University of Texas Health Science Center at Houston