Blood-clotting protein may be new target for Alzheimer's drugs
Despite the rapid rise of Alzheimer’s disease — the Alzheimer’s Association predicts as many as 7.7 million cases by 2030 — there are no preventative treatments available, few in the pharmaceutical pipeline, and those drugs being developed all share the same two molecular targets. Now Rockefeller University researchers report that by targeting a different molecule, a blood-clotting protein called fibrin, they could reduce inflammation in the brains of mice with different models of the disease.
Even as people with Alzheimer’s disease begin to lose bits of themselves, their brains are gathering material. Misfolded amyloid-β proteins build up, coalescing into dense plaques; another misformed protein, tau, aggregates inside neurons and causes visible neurofibrillary tangles. These two proteins have garnered the most attention and are at the center of most therapeutic research.
But in research published this month in the Journal of Experimental Medicine, graduate student Justin Paul and research professor Sidney Strickland, head of the Laboratory of Neurobiology and Genetics, show that a previously unexplored finding — that brains of Alzheimer’s patients have increased fibrin levels — could prove a potent method of attack.
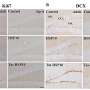
Scientists have known for years that Alzheimer’s damages the barrier that prevents blood from seeping into the brain, damage that lets fibrin and other blood proteins leak through. But it was a correlation that had gone largely unexamined. So Paul, Strickland and Jerry Melchor, a former postdoc in the lab, first looked at whether there was a correlation between fibrin and amyloid-β build-up in the brains of three different mouse models of Alzheimer’s. Sure enough, as amyloid-β accumulated, so did fibrin.
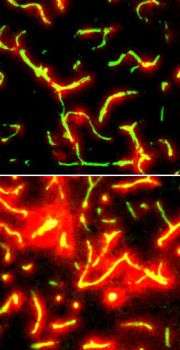
The researchers then looked to see if they could manipulate fibrin deposition in the brain. To decrease it, they used an enzyme from snake venom called ancrod, which causes the liver to clear fibrin from the bloodstream. To increase fibrin they chose tranexamic acid, as it effectively prevents the breakdown of fibrin clots. Both chemicals had their predicted effects, but closer examination revealed another noteworthy result: Although the amyloid-β plaques in the mice’s brains did not change with fluctuating fibrin levels, the microscopic inflammatory cells surrounding them did. “The number of microglia associated with each plaque — the number of the little inflammatory cells that get excited by fibrin deposition — are reduced when there’s less fibrin around, and increased when you can’t degrade the fibrin,” Paul says.
Not only that, but decreasing fibrin levels with ancrod also reduced the blood-vessel deterioration in the brain. “So reducing inflammation might be the link to preventing blood-brain barrier damage, as well,” he says.
Although Paul has not yet determined whether treating a pre-Alzheimer’s mouse with ancrod might prevent amyloid-β plaques from forming at all, even preventing inflammation would be a huge advance. “Research has shown a link between inflammation and cognitive deficits,” he says. “Fibrin is a critical component for increased inflammation in Alzheimer’s disease. And fibrin and fibrinogen, based on our study, should be considered a new therapeutic target. Although amyloid-β may be the cause of Alzheimer’s, alternative approaches need to be considered.”
Citation: Journal of Experimental Medicine 204(8): 1999-2008 (August 6, 2007)
Source: Rockefeller University