Molecules work the day shift to protect the liver from accumulating fat (w/ Video)
The liver normally makes and stores fat, which is required in moderation for normal body function. However, if the process goes awry, excess fat in the liver can cause major liver damage. In fact, fatty liver is a leading cause of liver failure in the United States, and is often brought on by obesity and diabetes. In turn, the increasing prevalence of these diseases has brought with it an epidemic of liver disease.
Abnormal sleep patterns, such as those of shift-workers, can be risk factors for obesity and diabetes. Investigators have known for decades that fat production by the liver runs on a 24-hour cycle, the circadian rhythm, and is similar to the sleep-wake cycle. A research team led by Mitchell Lazar, MD, PhD, director of the Institute for Diabetes, Obesity, and Metabolism at the University of Pennsylvania School of Medicine, has discovered molecules that act as "shift workers" to maintain the daily rhythm of fat metabolism. When those molecules do not do their jobs, the liver dramatically fills with fat. These findings are reported in this week's issue of Science.
Lazar and his colleagues, including Cell and Molecular Biology graduate student Dan Feng, found a team of molecules that, in normal mice, migrates to the genome of liver cells during the daytime. One of the team members, a protein called Rev-erb, delivers the molecular workers to thousands of specific locations in the liver genome, many of which are near genes involved in the production of fat. Another team member, called histone deacetylase 3 (HDAC3), does construction work on the protein scaffold (the epigenome) surrounding the genome to dampen the activity of the fat-related genes.
"This work shows that the epigenome, which is critical for regulating how genes are expressed, undergoes reversible remodeling every day", said Lazar. "This leads to a circadian rhythm of metabolism that is important, because disruption of this rhythm leads to fatty liver. This may explain in part why altered circadian rhythms in people who do shift work is associated with metabolic disorders."
Histones are proteins found in the nucleus that package and order DNA into structural units. Changes to these epigenetic structures alter how DNA folds in chromosomes, making genes less or more accessible to regulatory proteins and enzymes that copy genes into RNA messages.
Construction Team
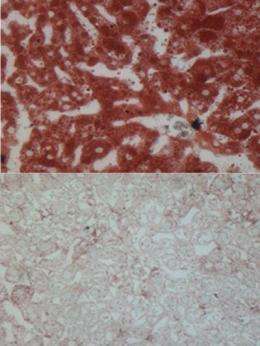
During the night, the day shift molecules depart the liver genome, and fat production increases due to other regulatory molecules. The fat production is kept in check when the Rev-erb construction team returns to the genome the next day. However, if either Rev-erb or HDAC3 is prevented from doing its job, the cycles do not occur, and the liver fills with fat.
By sequencing the DNA associated with HDAC-3 in the liver the Penn team found HDAC in 100 places in the liver genome at 5:00am, but 12 hours later at 5:00pm, HDAC was present in 15,000 places in the liver genome, indicating that it had been brought to the liver during the day. They also found that Rev-erb follows the same daily pattern, because it is the protein that gives HDAC-3 a ride to work.
The Lazar lab is looking in other tissues – fat cells, muscle, for example – to see if the same team of molecules is at work, as well as delving deeper into human applications to see how the findings may help explain what goes wrong with fat production and storage in conditions such as metabolic syndrome, insulin resistance, and diabetes. These findings also raise the interesting question of whether certain drugs should be given at specific times of day, to have greater benefit with reduced side-effects.
















