Can a cholesterol drug prevent colon cancer?
Thomas Jefferson University has started recruiting patients for a new National Cancer Institute (NCI)-sponsored clinical trial to test whether the cholesterol-reducing drug rosuvastatin is effective in the prevention of recurrent colon cancer.
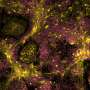
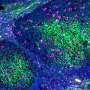
Previous laboratory research and population studies have shown that patients taking statins, the class of drugs that lowers cholesterol, had fewer colon polyps, which can lead to cancer if left untreated. However, those findings come largely from retrospective, observational studies originally designed to investigate lipid-lowering or cardiovascular endpoints in the short term rather than tumor endpoints.
"The jury is still out, and we need to get definitive answers," said Bruce Boman, M.D., Ph.D, professor of medical oncology at Thomas Jefferson University and principal investigator for the national clinical trial. "This prospective design comparing a statin against a placebo is what is needed to address the question: Are statins effective chemoprevention agents or not?"
This five-year, nationwide study will be the first randomized, prospective, placebo-controlled, double-blind study to evaluate the drug's role in preventing colon cancer and will involve 1,740 patients in total.
Conducted by a network of cancer research professionals from the National Surgical Adjuvant Breast and Bowel Project (NSABP) at 400 medical centers across North America, including Jefferson, the study involves patients who have recently been diagnosed with early stage colon cancer, and who were not already taking statins for high cholesterol. Those recruited have been surgically treated for stage I and II colon cancers previously.
Patients will be randomly assigned to one of two groups. Each group will take one pill a day for five years. One group will receive rosuvastatin, while the other group will receive a placebo.
The cumulative incidence for developing colorectal adenomas three years after surgery/treatment for early stage colon cancer is 50 percent. Thus, the benefit/risk ratio for chemoprevention intervention is potentially very positive in this high-risk population.
"There will be an estimated 102,900 new cases of colon cancer in the United States this year," said Norman Wolmark, M.D., NSABP's chairman. "In fact, colorectal cancer is the third most common cancer found in men and women in this country. We hope this trial will be an important step in reducing these numbers."
The principal investigator for the trial at Jefferson is Scott Goldstein, M.D, director of the Division of Colon and Rectal Surgery at Thomas Jefferson University.