Novel therapy for metastatic kidney cancer developed
Researchers at Virginia Commonwealth University Massey Cancer Center and the VCU Institute of Molecular Medicine (VIMM) have developed a novel virus-based gene therapy for renal cell carcinoma that has been shown to kill cancer cells not only at the primary tumor site but also in distant tumors not directly infected by the virus. Renal cell carcinoma is the most common form of kidney cancer in adults and currently there is no effective treatment for the disease once it has spread outside of the kidney.
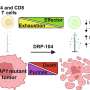
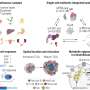
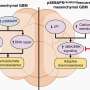
The study, published in the journal Cancer Biology & Therapy, tested Sorafenib (Nexavar), a drug approved by the FDA to treat kidney cancer, in conjunction with a novel adenovirus (Ad.5/3-mda-7). Adenoviruses are viruses known to infect the upper respiratory tract. But when used for therapeutic purposes, the virus' harmful genetic material is replaced with genetic code that activates biological processes within infected cells. The Ad.5/3-mda-7 adenovirus used in this study was engineered to cause kidney cancer cells and normal cells protecting the kidneys to express the cancer-killing protein MDA-7/IL-24. The therapy in this study was developed by Paul Dent, Ph.D., Universal Corporation distinguished professor in cancer cell signaling at VCU Massey and vice chair of the department of neurosurgery; and Paul B. Fisher, M.Ph., Ph.D., Thelma Newmeyer Corman endowed chair in cancer research at VCU Massey, professor and chair of the department of human and molecular genetics and director of VIMM; in collaboration with David T. Curiel, M.D., Ph.D., from the University of Alabama.
"While further research is needed, this therapy could be a novel and effective way to treat metastatic kidney cancer and prolong patient survival," says Dent. "This is the first study to clearly define that gene therapeutic delivery of MDA-7/IL-24 in kidney cancer should be explored in the clinic, especially since we've demonstrated an established, FDA-approved drug enhances its toxicity to cancer cells."
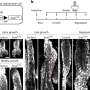
In mouse models, injection of the virus caused kidney cancer cells and normal cells lining the kidneys to secrete the protein MDA-7/IL-24. In the primary tumor site where the virus was first injected, the secreted MDA-7/IL-24 protein caused the tumor to stop growing. And once the protein entered the blood stream it stopped the growth of a second, distinct tumor not directly infected by the adenovirus, a result called a "toxic bystander effect." Normal cells were unaffected. Sorafenib, already approved by the FDA to treat renal carcinoma, enhanced MDA-7/IL-24 toxicity in the laboratory and significantly increased its anti-tumor effects in animal tumor models.
"Adenoviral gene therapies are still very new, but they represent a potentially powerful tool in the fight against cancer," says Fisher. "Our ultimate goal is to move our research from the laboratory to patients. And based on our findings, we hope these therapies will be effective against a variety of cancers."
The MDA-7/IL-24 gene was originally discovered by Fisher when he was a professor at Columbia University College of Physicians and Surgeons in New York. The current research was developed by Dent and Fisher at VCU Massey Cancer Center and the VIMM, and the method by which the virus infects the cancer cells was developed in collaboration with Curiel. The investigators hope to advance the research to a Phase I clinical trial on patients with metastatic kidney cancer, and also are investigating the use of MDA-7/IL-24 gene therapy on other diseases, including melanoma and brain, prostate, pancreatic, breast and colon cancers.