A chemical to make brain cells grow: Mental decline thwarted in aging rats
Scientists have discovered a compound that restores the capacity to form new memories in aging rats, likely by improving the survival of newborn neurons in the brain's memory hub. The research, funded in part by the National Institutes of Health, has turned up clues to a neuroprotective mechanism that could lead to a treatment for Alzheimer's disease.
"This neuroprotective compound, called P7C3, holds special promise because of its medication-friendly properties," explained Steven McKnight, Ph.D., who co-led the research with Andrew Pieper, M.D., Ph.D., both of University of Texas Southwestern Medical Center, Dallas. "It can be taken orally, crosses the blood-brain barrier with long-lasting effects, and is safely tolerated by mice during many stages of development."
The researchers report on their findings July 9, 2010 in the journal Cell. Their work was funded, in part, by the NIH's National Institute of Mental Health (NIMH), a NIH Director's Pioneer Award funded through the Common Fund and managed by the National Institute of General Medical Sciences, and National Cancer Institute.
"This striking demonstration of a treatment that stems age-related cognitive decline in living animals points the way to potential development of the first cures that will address the core illness process in Alzheimer's disease," said NIMH Director Thomas Insel, M.D.
Physical activity, social, or other enriching experiences promote neurogenesis - the birth and maturation of new neurons. This growth takes place in the dentate gyrus, a key area of the brain's memory hub, the hippocampus. But even in the normal adult brain, most of these newborn neurons die during the month it takes to develop and get wired into brain circuitry. To survive, the cells must run a gauntlet of challenges. Newborn hippocampus neurons fare much worse in aging-related disorders like Alzheimer's, marked by runaway cell death.
In hopes of finding compounds that might protect such vulnerable neurons during this process, Pieper, McKnight and colleagues tested more than 1000 small molecules in living mice. One of the compounds, designated P7C3, corrected deficits in the brains of adult mice engineered to lack a gene required for the survival of newborn neurons in the hippocampus. Giving P7C3 to the mice reduced programmed death of newborn cells - normalizing stunted growth of branch-like neuronal extensions and thickening an abnormally thin layer of cells by 40 percent. Among clues to the mechanism by which P7C3 works, the researchers discovered that it protects the integrity of machinery for maintaining a cell's energy level.
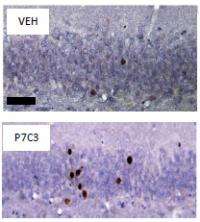
To find out if P7C3 could similarly stem aging-associated neuronal death and cognitive decline, the researchers gave the compound to aged rats. Rodents treated with P7C3 for two months significantly outperformed their placebo-treated peers on a water maze task, a standard assay of hippocampus-dependent learning. This was traced to a threefold higher-than-normal level of newborn neurons in the dentate gyrus of the treated animals. Rats were used instead of mice for this phase of the study because the genetically engineered mice could not swim.
The researchers pinpointed a derivative of P7C3, called A20, which is even more protective than the parent compound. They also produced evidence suggesting that two other neuroprotective compounds eyed as possible Alzheimer's cures may work through the same mechanism as P7C3. The A20 derivative proved 300 times more potent than one of these compounds currently in clinical trials for Alzheimer's disease. This suggested that even more potent neuroprotective agents could potentially be discovered using the same methods. Following up on these leads, the researchers are now searching for the molecular target of P7C3 - key to discovering the underlying neuroprotective mechanism.
More information: References:
-- Discovery of a Pro-neurogenic, Neuroprotective Chemical. Pieper AA, Xie S, Capota E, Estill SJ, Zhong J, Long JM, Becker GL, Huntington P, Goldman SE, Shen CH, Capota M, Britt JK, Kotti T, Ure K, Brat DJ, Williams NS, MacMillan KS, Naidoo J, Melito L, Hsieh J, Brabander JD, Ready JM, McKnight SL. 2010, Jul 8. Cell, DOI:10.1016/j.cell.2010.06.018
-- A selective allosteric potentiator of the M1 muscarinic acetylcholine receptor increases activity of medial prefrontal cortical neurons and restores impairments in reversal learning. Shirey JK, Brady AE, Jones PJ, Davis AA, Bridges TM, Kennedy JP, Jadhav SB, Menon UN, Xiang Z, Watson ML, Christian EP, Doherty JJ, Quirk MC, Snyder DH, Lah JJ, Levey AI, Nicolle MM, Lindsley CW, Conn PJ. J Neurosci. 2009 Nov 11;29(45):14271-86.PMID: 19906975
-- Discovery and Characterization of Novel Subtype-Selective Allosteric Agonists for the Investigation of M1 Receptor Function in the Central Nervous System. Lebois EB, Bridges TM, Lewis LM, Dawson ES, Kane AS, Xiang S, Jadhav SB, Yin H, Kennedy JP, Meiler J, Niswender CM, Jones CK, Conn PJ, Weaver DC and Lindsley CW. ACS Chem. Neurosci., 2010, 1 (2), pp 104-121, September 25, 2009. DOI: 10.1021/cn900003h















