'Achilles' heel' in Y chromosome linked to sex disorders

The unique mechanism behind the evolutionary survival of the human Y chromosome may also be responsible for a range of sex disorders, from failed sperm production to sex reversal to Turner Syndrome.
Roughly six years ago, David Page's lab at Whitehead Institute for Biomedical Research reported the discovery of eight large areas of mirror-imaged genetic sequences, or palindromes, along the Y chromosome. Because the Y chromosome essentially has no partner with which to swap genes, a process that between ordinary chromosome pairs leads to genetic diversity and the exchange of good genes for damaged ones, it relies on its own palindromes to swap genes with itself. The Y, as it turns out, folds itself in the middle of palindromic regions, thereby pairing identical sequences to allow for potentially beneficial genetic exchange.
At the time, the finding provided explanation for why, despite much-heralded reports to the contrary, the Y chromosome is not doomed to extinction. Now, the Page lab has discovered that the Y's process of self-preservation can randomly go awry, with considerable clinical consequence.
"This is the sequel to the Y chromosome palindrome story," says Page, Whitehead Institute Director and a Howard Hughes Medical Institute investigator.
The latest chapter of the story, whose conclusion is published in the September 4 issue of Cell, began with the intriguing hypothesis that perhaps the Y's process of self-recombination can inadvertently turn the entire chromosome into a palindrome—literally, a mirror-image of itself. The result would be a so-called isodicentric Y chromosome (idicY), an abnormal structure with, as the name implies, two centromeres.
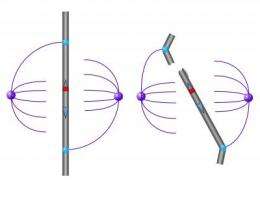
"We began to think seriously about the centromeres and the activity around them. Two centromeres render the chromosome susceptible to damage," says Julian Lange, first author of the Cell paper and a former graduate student in the Page lab. Because of the Y chromosome's well known roles in sex determination and male fertility, Lange began to speculate about the potential clinical impact of the transmission of an idicY during fertilization.
"Because the Y chromosome is not essential to an individual's survival, these isodicentric Ys can persist," says Lange, who, after completing this research at Whitehead, became a postdoctoral fellow at Memorial Sloan-Kettering Cancer Center. "They can be found in the population."
And Lange found them, in the DNA samples of 51 patients screened from a field of nearly 2400 individuals who had come under study over the course of many years because of failed sperm production, structurally abnormal Y chromosomes, or sex reversal. Through sophisticated genetic analysis, it became clear that idicYs were responsible for spermatogenic failure in many of the male patients.
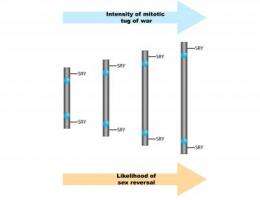
However, 18 of the 51 patients were anatomically female—despite having two copies of the male-determining SRY gene on their idicY chromosomes. Lange and Page began to hypothesize that the feminization they had identified was related to the instability of the idicYs themselves. Suspecting that the level of instability of an idicY increases with the size of the chromosome, they looked for a connection and found something somewhat paradoxical: the larger the Y chromosome, the greater the likelihood of sex reversal.
"We had predicted this correlation, which relates to the overall distance between the centromeres," says Page. "But when we confirmed it with the patient data, we were blown away."
Page believes that this new model for the formation of idicY chromosomes, coupled with the size-instability correlation, suggests a causal link to Turner syndrome, a chromosomal abnormality in girls or women, characterized by the lack of one sex chromosome. Turner syndrome affects an estimated 1 in 2500 females. Page won't yet speculate as to what percentage of Turner syndrome could be caused by this palindrome-to-palindrome recombination, but he does think it's significant.
Source: Whitehead Institute for Biomedical Research
















