IDO2 an active enzyme to target in pancreatic cancer
An enzyme that is overexpressed in pancreatic cancer cells may hold the key to successfully treating the disease with targeted immunotherapy, researchers from Thomas Jefferson University reported at the 2008 Annual Meeting of the Southern Surgical Association.
Previous data show that a protein, indoleamine 2,3-dioxygenase (IDO), is overexpressed in pancreatic ductal adenocarcinomas, according to Jonathan R. Brody, Ph.D., an assistant professor in the Department of Surgery at Jefferson Medical College of Thomas Jefferson University in Philadelphia, and co-director of the Jefferson Center for Pancreas, Biliary and Related Cancers. The center is led by Charles J. Yeo, M.D., Samuel D. Gross Professor and chair of the Department of Surgery, who was also involved with the study.
According to Dr. Brody, IDO is an enzyme that represses the immune system, thus protecting the cancer cells and helping them evade immune detection. The Jefferson researchers and their collaborators from the Lankenau Institute for Medical Research (LIMR) in Wynnewood, Pa., previously reported that the IDO inhibitor D-1-methyl-tryptophan (1-MT), preferentially targets a related protein, IDO2.
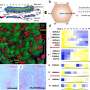
"Our data are the first that report expression of the IDO2 protein in malignant pancreatic tissue," Dr. Brody said. "About 75 percent of the patients in our cohort have an active enzyme based on genetic analysis. This puts forth the concept of genotyping patients for the IDO2 enzyme, to identify patients who may respond to a therapeutic strategy including an IDO inhibitor, a potential novel drug for pancreatic cancer."
Dr. Brody and his team, which included George Prendergast, Ph.D., and Richard Metz, Ph.D., from LIMR, sequenced IDO2 in 36 resected pancreatic ductal adenocarcinomas to evaluate how many patients harbor an active enzyme in relation to well-known polymorphisms: R235W and Y359STOP. Presumably, only one functional allele needs to be present in order to have an active IDO2 enzyme.
Nineteen percent of the patients were homozygous wild-type, meaning they had two active alleles, and 42 percent of the patients were heterozygous, meaning they had one active allele. Twenty-five percent were homozygous for two inactive IDO2 alleles. The remaining 14 percent were difficult to determine due to limits in sequencing and distinguishing the alleles, but were most likely heterozygous, according to Dr. Brody.
Phase-1 studies of 1-MT, the IDO inhibitor are currently underway.
Source: Thomas Jefferson University