Study provides insight on a common heart rhythm disorder
University of Iowa researchers and colleagues in France have identified a gene variant that causes a potentially fatal human heart rhythm disorder called sinus node disease. Also known as "sick sinus syndrome," the disease affects approximately one in 600 heart patients older than 65 and is responsible for 50 percent or more of the permanent pacemaker placements in the United States.
While the newly discovered gene variant is rare, the study provides insight into cellular mechanisms that regulate sinus node function and identifies an unanticipated new pathway for developing future therapies to regulate more common forms of sinus node disease. The findings, which also have research implications beyond heart disease, were published online Oct. 1 by the Proceedings of the National Academy of Sciences.
The team first analyzed data from two families in France: a family of 74 individuals, 26 of whom had sinus node dysfunction, and a family of 44 individuals, 13 of whom had the disease. Many of the affected individuals carried the same gene variant, and many experienced variable heart rate and bradycardia (dangerously low heart rate).
The investigators found that variants in a gene called ankyrin 2, or ANK2, resulted in dysfunction in the protein ankyrin-B in the members of these two different families, said the study's senior author Peter Mohler, Ph.D., associate professor of internal medicine in the University of Iowa Carver College of Medicine.
"While a small number of the patients displayed heart disease symptoms, including ventricular arrhythmias, the prevalence of sinus node dysfunction in these patients was extremely high. In fact, most required the implantation of cardiac pacemakers," said Mohler, who also is a Pew Scholar. "We predict that there are likely additional unidentified ankyrin variants in the larger general population that predispose humans to a combination of heart disease symptoms, including sinus node dysfunction, atrial fibrillation and ventricular arrhythmias.
"We were fortunate in this study to assemble an amazing collaboration between outstanding basic science laboratories at the University of Iowa and a group of internationally-renowned clinical electrophysiologists and geneticists in France," he added.
Blood circulation through the body depends on the coordinated contraction of specialized heart cells called ventricular cardiomyocytes. Cells in the sinoatrial node have a different role -- to maintain the heart's normal rhythm.
"There are thousands of ventricular cells in the heart, but there are only hundreds of sinoatrial node cells, so each nodal cell is absolutely critical for every beat of the human heart," Mohler said.
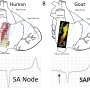
Because there are so few sinoatrial node cells, it has been difficult until recently for scientists to get sample cells from human or animal models, noted Thomas Hund, Ph.D., a member of the study team and University of Iowa associate in internal medicine.
"Building on others' discoveries, scientists in Professor Mohler's Professor Mark Anderson's, and Assistant Professor Long-Sheng Song's lab perfected methods to isolate mouse sinoatrial node cells. We were then able to image these cells and see what was different about them," Hund said.
Based on research previously published by Mohler in 2003 in the journal Nature, the investigators knew that in ventricular cardiomyocytes the protein ankyrin-B behaves like a "tugboat," delivering ion channels and anchoring them to specific domains on the cell membrane. These ion channels serve as conduits for the electrical activity that triggers cell contraction. The team found that ankyrin plays a similar critical role in organizing ion channels for the sinoatrial node cells, making it possible for these cells to maintain the normal heart rhythm.
"Taken together, these studies suggest that ankyrin-B and similar proteins organize cell systems to ensure that cellular components don't float around randomly," Mohler said. "Understanding how these cells behave may help us learn how to fix malfunctions that occur when people age or experience common forms of cardiac disease.
"Genes that make ankyrin or proteins like it are probably going to be the next set of genes targeted for understanding diseases that involve cells with electrical activity," he added. "In addition to heart disease, ankyrins could be involved in other 'excitable' cell diseases such as epilepsy, bipolar disease and diabetes."
Source: University of Iowa