'Stuffy nose' mouse: A promise to help treat 31 million with sinusitis
Mice with inflamed nasal tissue being tested at a Johns Hopkins laboratory may be unable to tell if something smells bad or good, but their sensory deficit is nothing to turn up a nose at.
That is because, their developers say, the mice's reversible loss of one of their key senses, which is essential to tasting food or sensing danger from foul odors, sets them apart from all other mice and binds them to an estimated 31 million Americans living with chronic sinusitis, a persistent inflammation of the tissue that lines the nasal and sinus cavities. Add to this group, millions of people with other disorders that affect smell, including viral infections, head traumas, tumors, Alzheimer's and Parkinson's diseases.
"A sense of smell in good working order is essential to our quality of life, and these genetically engineered mice give us the first real animal model for better understanding, treating and preventing people from suffering a loss of olfactory function due to sinonasal inflammation," says sinusitis expert Andrew Lane, M.D., who led the team that developed the olfactory-compromised mice.
"And because we can turn on and off the inflammation in these mice, we really can mimic how the most overlooked and very disabling aspect of sinusitis, the loss of smell, or anosmia, plays out in people," says Lane, an associate professor at the Johns Hopkins University School of Medicine.
Lane will cite smell and sinus tissue data from his studies with mice, and he will compare them to other clinical data, when he introduces the inflammation-induced, anosmic mice to fellow experts July 22 during a presentation at the XV International Symposium on Olfaction and Taste, in San Francisco, Calif.
"Until now, the lack of realistic animal models for each of the key symptoms of chronic inflammation in the nasal tissue - such as the growth of nasal polyps, the loss of the sense of smell, swollen sinus tissue, or clogged and runny noses - has slowed sinusitis research and hindered our search for therapies," says Lane, director of the Johns Hopkins sinus center, where he treats hundreds of patients with the condition.
New therapies are needed, he says, as an alternative to long-term steroids, which block the inflammatory chemical pathway but also have debilitating side effects, including loss of bone density, cataracts in the eye and weight gain.
Another key advantage to the new sinusitis mouse, he points out, is that it can be more easily studied than human olfactory tissue, which is surgically difficult to cut out from deep inside the skull and because the tissue sits dangerously close to the brain.
Johns Hopkins scientists began their quest for a "stuffy nose" mouse with inflammation-produced anosmia in 2002.
Their first steps were performed in the lab, where researchers genetically modified developing mouse cells to breed a family that could secrete key cytokine proteins only in the olfactory, uppermost part of the nose. An overproduction of cytokines, which are better known for their role in the body's immune response to disease-causing pathogens, are a telltale chemical signature in sinusitis.
Lane's team focused its efforts on one of hundreds of cytokines, specifically, tumor necrosis factor alpha, or TNFα, because of its many links to sinusitis. TNFα is overactive during all kinds of inflammation, and the chemical is also known to accelerate olfactory nerve cell turnover. Unlike most other kinds of nervous tissue, the olfactory type can grow back, an evolutionary adaptation to the constant shedding of skin cells that line the nasal cavity.
Researchers first injected mouse egg cells with a gene for TNFα and a control system so that cells with the gene would secrete the cytokine on demand and only if activated.
In a second set of mice, Lane's team planned to activate the control system only in olfactory tissue, by genetically implanting the controls to another gene, called CYP2G1, which is produced only in the mouse nose, specifically in its nourishing sustentacular cells that sit between nasal nerve cells.
Lane says the system had to be "nasally specific," so that secretion of TNFα occurred in the mouse, much like it does in sinusitis in humans.
After breeding the two groups of mice to get their animal test model, of which there are 20 at any given time, scientists then turned on TNFα production by stimulating the sustentacular cells with tetracycline, an antibiotic trigger that was added to the mice's drinking water. The system remained off when no tetracycline was added.
To make sure the model worked, mice were fed the drugged drinking water for nearly two months, and samples of olfactory tissue were tested weekly for any sense of smell in response to various odors.
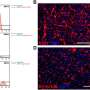
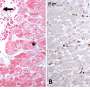
Results showed that sense of smell, as gauged by minute electrical currents in olfactory tissue, dropped progressively, by half (50 percent) within two weeks, and stopping completely after six. When tissue was viewed under microscope, white blood cells were visible, a telltale sign of inflammation. Olfactory nerve cells had nearly disappeared.
But when researchers stopped the drug-induced sinusitis, olfactory nerve cells rebounded and grew back within a couple of weeks, "proving that what we have is a mouse with reversible olfactory loss due to inflammation, which should speed up our learning more about the disease and testing new therapies," says Lane. "Ultimately, we hope to develop treatments that allow the sense of smell to recover, even in the presence of a hostile inflammatory environment due to sinusitis."
His team's next steps will be to test different cytokines, either alone or in combination, to clarify their roles in the loss of smell in sinonasal inflammation.
Future studies are also planned to monitor the effects of current steroid therapies on mouse olfactory tissue, in the hope of modifying or bolstering the treatments and speeding up delivery of these medications to inflamed tissue.
Another phase of research, he says, involves testing other anti-inflammatory drugs, such as infliximab (Remicade), which is used to treat arthritis, to see if they can spur growth of olfactory neurons during sinusitis.
Lane also plans to add more sinusitis features to the animal model, including progressive swelling of sinus tissue and rhinitis.
Source: Johns Hopkins Medical Institutions