Researchers discover how HIV turns food-poisoning into lethal infection
Nearly half of all HIV-positive African adults who become infected with Salmonella die from what otherwise would be a seven-day bout of diarrhea. Now, UC Davis School of Medicine scientists have discovered how salmonella becomes lethal for AIDS patients. Their findings also implicate a mechanism by which HIV evades the powerful drugs used to treat AIDS.
“We have found the defect in the immune response that allows Salmonella to cross the mucosal barrier of the gut, enter the bloodstream and infect other organs,” said Andreas Bäumler, a UC Davis professor of medical microbiology and immunology and co-author of the study.
The results of the study, which will be published online by Nature Medicine March 23, revealed that viral infection of the intestine results in the depletion of a type of white blood cell, called Th-17, in the gut mucosa. This T helper lymphocyte produces IL-17, a cytokine or chemical messenger that plays a crucial role in the inflammatory response, recruiting other immune system cells to the site of infection.
This kind of interruption in the gut’s immune response could be allowing HIV to maintain reservoirs that evade drug treatments, said Satya Dandekar, professor and chair of the department of medical microbiology and immunology.
“It’s like putting out the fire, but leaving the embers smoldering,” Dandekar said.
The rise in patients with acquired immune deficiency syndrome (AIDS) in sub-Saharan Africa has led to a dramatic increase in the frequency of non-typhoidal Salmonella serotypes (NTS), the strains of the bacteria that cause acute food-borne disease world wide. Normally, this infection is limited to the intestine, causing gastroenteritis. In AIDS patients, however, the infection spreads to the bloodstream and causes what is called NTS bacteremia.
While at a conference, Bäumler was surprised to learn from epidemiologist and physician Melita Gordon of the University of Liverpool that Salmonella was quickly becoming one of the leading causes of death in parts of Africa. (Gordon is a co-author on the current paper.) Bäumler returned to Davis and approached Dandekar about collaborating.
Dandekar had been studying the role of gut-associated lymphoid tissue in HIV. In a 2006 study, she found that HIV continued to replicate in the gut mucosa and suppress immune function in patients being treated with antiretroviral therapy — even when T-cell counts from blood samples from the same individuals indicated antiretroviral treatment was working.
“We think the real battle between an individual’s immune system and HIV is happening in the gut mucosa where there is massive destruction of immune cells,” Dandekar said. Gut-associated lymphoid tissue, she pointed out, accounts for 70 percent of the body’s immune system.
In HIV-infected patients, there is a gradual loss of CD4+ T cells over time. These cells, also called T helper cells, organize the immune system’s attack on disease-causing invaders, like Salmonella. Unlike the steady decline of T cells in peripheral blood, there is a very rapid loss of CD4+ T cells in the gut mucosa, Dandekar said.
“We wanted to know whether the loss of the CD4+ T-cells in the gut contributed to the inactivation of the immune system one sees in HIV-infected patients,” she said.
Both Bäumler and Dandekar said the timing was perfect for their collaboration. Together, they developed a novel technique that allowed them to study early intestinal responses to Salmonella infection in rhesus macaques infected with simian immunodeficiency virus (SIV), an established model for HIV infection.
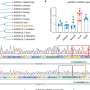
“We found that animals that had no SIV infection were able to generate immediate responses to bacterial exposure, producing Th17 cells in large amounts,” Dandekar said. The SIV-infected animals, however, had either a significantly lower response or lacked did not produce measurable amounts of the cytokine.
“This muted Th17 response led to dissemination of Salmonella from the gut to the peripheral blood,” Dandekar said.
The team of researchers also used mice that lacked the IL-17 receptor, an arm of the mucosal immune response, to confirm that IL-17 deficiency leads to increased systemic dissemination of Salmonella.
“We believe IL-17 deficiency causes defects in the mucosal barrier of the gut,” Dandekar said.
Both Bäumler and Dandekar agreed that the results of their collaboration have exciting implications for both HIV and Salmonella research and, more importantly, get scientists closer to finding treatments for HIV and the deadly form of Salmonella.
In terms of HIV, the results suggest that Th17 may make a good biomarker for monitoring HIV infection and testing the efficacy of vaccines and other therapies. They also suggest that efforts to enhance Th17 function may improve existing antiretroviral treatments.
“We are interested in looking at different molecules and compounds to see if we can boost mucosal immune defenses in the gut,” she said.
Dandekar is also interested in looking at Th17 function in those who respond well to treatment and in long-term non-progressors, those individuals who carry HIV for years without going onto develop AIDS.
“Now we know these cells are playing a big role, but we need to better understand how they are contributing to immune inactivation and inflammation,” Dandekar said.
In terms of Salmonella, Bäumler’s next step is to discover the mechanisms by which non-immunocompromised patients are able to rid themselves of the infections.
“We now know which cytokines orchestrate the mucosal barrier function, but we still don’t know what kills these bacteria,” he said.
Source: University of California - Davis