Human C-reactive protein regulates myeloma tumor cell growth and survival
Scientists report that a protein best known as a common marker of inflammation plays a key role in the progression of human cancer. The research, published by Cell Press in the September issue of the journal Cancer Cell, implicates C-reactive protein (CRP) as a potential target for cancer treatment.
CRP is a protein that is made in the liver and secreted systemically during the process of inflammation in response to the inflammatory cytokine IL-6. The blood level of CRP is elevated in patients with infections, inflammatory diseases, some cardiovascular diseases, and malignancies including multiple myeloma (MM). Dr. Qing Yi and Dr. Jing Yang from the M.D. Anderson Cancer Center and colleagues designed a series of studies to investigate whether human CRP might influence the growth and survival of MM tumor cells.
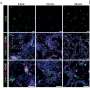
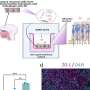
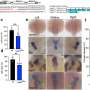
The researchers found that addition of CRP to cell cultures at levels observed in patients with MM promoted myeloma cell proliferation and protected myeloma cells from chemotherapy-induced apoptosis and apoptosis induced by IL-6 withdrawal. The protective influence of CRP was also validated in a mouse model of myeloma.
The researchers went on to investigate the cell signaling pathways underlying CRP-mediated protection of myeloma cells. They demonstrated that CRP enhanced secretion of IL-6; binds activating Fcg receptors; activates PI3K/Akt, ERK, and NF-kB pathways; and inhibits caspase cascade activation induced by chemotherapy drugs. Further, CRP was shown to synergize with IL-6 in protecting myeloma cells from apoptosis.
These results provide strong evidence that CRP is not just a marker for MM but is a critical regulator of myeloma cell survival. “CRP protects myeloma cells from apoptosis induced by chemotherapy drugs and stimulates myeloma cells to secrets more IL-6, which in turn provides additional protection to myeloma from apoptosis and stimulates liver cells to secrete more CRP. Thus, CRP could be a therapeutic target for breaking the vicious circle of myeloma to improve the therapeutic efficacy of currently available treatments,” explains Dr. Yi.
Source: Cell Press