'Lucky 13' as new gene discovery offers further hope for childhood blindness
An international research team has discovered a gene that, when mutated, causes one of the most common forms of inherited blindness in babies. Scientists at the University of Leeds, working in collaboration with experts from other centres around the world, identified the gene, which is essential to photoreceptors in the eye, the cells that "see" light.
The finding, the thirteenth gene to be linked to Leber’s congenital amaurosis (LCA), comes at a time of hope for the people born with the disorder. Scientists at Moorfields Eye Hospital, London, recently announced the start of clinical trials for a gene therapy involving injecting genes into the eye of patients with LCA to restore their sight. The finding of the new LCA gene, based on work funded by the Wellcome Trust and local charity Yorkshire Eye Research, appears in this month's edition of the journal Nature Genetics.
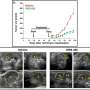
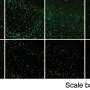
The newly-discovered gene, LCA5, is involved in the production of lebercilin, an essential component of photoreceptors in the retina. Lebercilin is found in other tissues as part of the cilia, finger-like projections from the surface of cells capable of moving molecules around. However, mutations in the LCA5 gene only appear to cause defects in the retina.
"We already know of a dozen genes which, when mutated, cause LCA," says Professor Chris Inglehearn from the Leeds Institute of Molecular Medicine at St James's Hospital, Leeds. "This new gene is the thirteenth and adds a substantial new piece to a growing body of evidence that defects of the cilia are a major cause of inherited blindness. In that sense, we can consider this a 'lucky thirteenth' as we are building a much clearer picture of what causes the disorder."
Professor Inglehearn believes that lebercilin may be involved in moving proteins from the inner to outer segments of photoreceptors in the retina. Protein transport is essential within retinal photoreceptor cells as they are long, thin cells with a highly evolved structure on one end (the outer segment) which detects light and sends signals to the brain, a function requiring a large amount of energy.
"LCA is usually a disease where protein function has been lost completely, but carriers of just one copy of the mutation, who will almost certainly have reduced protein levels, nevertheless function perfectly normally," explains Professor Inglehearn. "This being the case, restoration of even a tenth of the missing protein may be enough to restore vision. So our findings, together with the recently announced clinical trials, hold great promise."
Mutations in LCA5 are relatively rare. As it is a recessive gene, a child would need to be carrying two copies of the gene to develop LCA, one from each parent. However, the disorder is more common within populations where marriage to first or second cousins is common, such as the Pakistani community.
"If a parent is found to carry a mutation in the LCA5 gene, the risk of blindness in their children and grandchildren is still virtually zero as long as the other parent does not carry it," says Professor Inglehearn. "The odds of two parents both carrying the same or different mutations in the LCA5 gene is very low, but this increases where the parents are related."
Professor Inglehearn hopes that the findings will be useful to inform and counsel the families most at risk, particularly within the Pakistani communities both in the UK and Northern Pakistan.
The findings have been welcomed by Bruce Noble from Yorkshire Eye Research, which part-funded the research.
"Obviously we have to be careful how we interpret these results, as finding the genes doesn't automatically lead to a cure," says Mr Noble. "Nevertheless, this new result tells us something very important about what the eye is doing normally and about a new and common way in which it can go wrong. Given some exciting recent developments on testing possible cures for inherited blindness, its becoming very important for everybody to know exactly which mutation they've got, because the treatments being tested are specific for different kinds of retinal degeneration. All in all its an exciting time for eye research and a very promising one for people with these conditions. This new result is another important step in the right direction and Yorkshire Eye Research is very proud to have supported it."
The research was also welcomed by Professor John Marshall, Chairman of the Medical Advisory Board at the British Retinitis Pigmentosa Society.
"This dramatic discovery provides further information enabling us to combat blindness within the Retinitis Pigmentosa group," says Professor Marshall. "The more genes we discover the better we are placed to treat the diseases by methods such as that recently announced at Moorfields Eye Hospital – also supported by the BRPS."
Source: Wellcome Trust