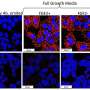
Researchers discover inherited mutation for leukemia
Researchers have discovered the first inherited gene mutation that increases a person's risk for chronic lymphocytic leukemia (CLL), one of the most common forms of the disease.
The study shows that the inherited mutation greatly reduces the gene's protective activity. Furthermore, a second kind of change occurs later that turns the gene off altogether, leading to leukemia. This latter alteration is a chemical change that is not inherited.
The findings could help identify people at risk for chronic leukemia, but they also may provide new insights into the process of natural cell death. They may even lead to new strategies for treating the disease.
The research is to be published in the June 1 issue of the journal Cell. It was led by researchers at the Ohio State University Comprehensive Cancer Center.
The mutation was found in a gene called DAPK1, which normally helps trigger the death of cells before they become cancerous. Researchers identified the mutation by testing a family in which the father, four sons, a grandson and a distant female relative developed this form of leukemia.
The chemical change is called DNA methylation. Healthy cells use this process to silence unneeded genes. But abnormal DNA methylation can turn off genes that control cell growth, and that lead to tumor growth.
"Our findings identify for the first time a gene that appears to be associated with hereditary CLL," says coauthor John C. Byrd, professor of internal medicine and a CLL specialist.
"They also show the importance of the gene in the pathogenesis of CLL, and direct us to target this gene with therapies that might re-activate it."
The findings also provide evidence that some genes might contribute to cancer even when they are not silenced entirely.
"This inherited change is remarkably subtle," says co-principal investigator Albert de la Chapelle, professor of molecular virology, immunology and medical genetics and a researcher with the Ohio State human cancer genetics program. "It does not shut down the gene, but just lowers its expression somewhat.
"Recently, many cancer geneticists have come to believe that such subtle changes are common causes of cancer, and this is one of the first, strong examples of that principle."
The study succeeded because it combined the field of gene mutation research and the new field of epigenetics, which identifies genes silenced by faulty DNA methylation, says Christoph Plass, professor of molecular virology, immunology and medical genetics and of veterinary biosciences, and also co-principal investigator on the study.
"Our findings show that it's important to look for both genetic and epigenetic alterations when identifying problem genes," he says.
CLL is the most common form of adult leukemia, with 15,300 new cases and 4,500 deaths from the disease expected this year in the United States. The leukemia is slightly more common in men than women, and typically strikes people who are in their 50s, 60s and 70s.
About 90 percent of CLL cases are sporadic; that is, they have no genetic component.
But about one in 10 people with CLL have relatives who also develop the disease – strong evidence of a hereditary predisposition. However, usually only two or three people within a family are affected, making it difficult to do the genetic studies needed to find possible mutations, Plass says.
The family examined in this research was identified by collaborator Henry Lynch at Creighton University.
The researchers are now studying the chemical pathway that regulates the gene, considering possible therapies, working to identify other CLL families and looking for other predisposing genes.
In 2005, other Ohio State Comprehensive Cancer Center researchers discovered a germline mutation in a gene for a microRNA that is implicated in CLL, suggesting that this may also be a predisposing mutation for the disease.
Source: Ohio State University