HIV protein acts as solvent, releasing viral particles from the surface of their host cell
In 1989, researchers discovered an HIV protein called Vpu that was key to how the AIDS virus spreads from cell to cell. Produced only by the HIV-1 virus and its closest relatives, Vpu appeared to be somehow involved in helping put together new viral particles and assisting with their release from the cell. Mutant viruses that lack the protein create infected cells with a distinctive characteristic: In these cells, the virus is made but isn’t released efficiently.
In the 17 years since the protein’s discovery, no clear theory has emerged to explain exactly how it works. But now, scientists at the Aaron Diamond AIDS Research Center (ADARC) and Rockefeller University have uncovered a very specific role for Vpu: It works like a solvent to “unstick” viruses from the membrane of the cell that produced them, allowing them to be released and to spread to adjacent cells. Research published this summer in PLoS Pathogens by Paul Bieniasz, associate professor at Rockefeller, explains this oddity for the first time. “We discovered that infected cells have a glue that sticks newly assembled virus particles to them, and prevents them from floating off to infect new cells,” Bieniasz says. “Vpu is like the anti-glue.”
A couple of years ago, a group in Tennessee discovered that primate cells don’t require Vpu for efficient virus release. Further research seemed to suggest that there was something in human, but not monkey, cells that was preventing virus release — something that the HIV virus could overcome by producing Vpu. “This type of thing is beginning to become a recurrent theme in HIV biology, the virus has evolved several activities to overcome inhibitors that cells make,” Bieniasz says.
Because cells infected with Vpu-deficient HIV are packed with virus-laden vesicles inside, as well as on their surface, it was an open question as to what function Vpu served. Were cells without Vpu sucking the particles back in? Or, as in a popular theory, in the absence of Vpu were the viruses being directed away from the cell surface and instead toward a cellular compartment called an endosome, which would then destroy them?
To figure out what was going on, Stuart Neil, the paper’s first author and a postdoctoral fellow in the Bieniasz lab, compared two types of human cells: HOS cells, in which Vpu doesn’t enhance virus release, and HeLa cells, in which it does. The results came as a surprise to the research team. When Neil blocked the activity of the endosomes in HeLa cells, he found that it decreased the accumulation of viral particles inside the cells – particles instead accumulated on the cell surface. But in HOS cells, where there was little intracellular accrual in the first place, blocking endosomal activity didn’t appear to do a thing. To Neil and Bieniasz, this suggested that the viral particles inside the cell were, indeed, derived from virus that had made it to the cell’s plasma membrane but then been pulled back into the interior. “So Vpu is doing something quite specific to virus release,” Neil says.
That something turned out to be acting like what Bieniasz refers to as “anti-glue.” When Neil added a protein-digesting enzyme to the Vpu-defective, virus-studded cells, the virus particles went drifting off into the surrounding medium. Their results imply that a cell’s surface is sticky to enveloped virus particles that have just pinched off. “And what Vpu appears to do is remove that protein glue,” Bieniasz says, “so the assembled particle can be released.” Their next step, he says, is figuring out exactly what the glue is.
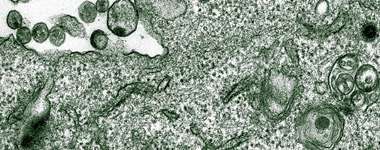
One of the especially surprising things about their results, Neil says, is that although older electron microscopy images of Vpu-defective viruses clearly show mature virus collecting at the cell surface as well as in endosomes, most studies didn’t use those images as a springboard and the observation remained unexplored. Now,the scientists' findings may also provide a glimpse at a larger story, one that has implications beyond HIV. This cellular glue is likely to be a problem for many other enveloped viruses as well, Bieniasz says. “We’re at the very early stages of studying this activity, but there are hints that other viruses have found different solutions to this problem.”
Source: Rockefeller University