This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Scientists map changes in soot particles emitted from wildfires
Not many people would voluntarily fly through plumes of smoke emitted from wildfires. But atmospheric scientists from the U.S. Department of Energy's Brookhaven National Laboratory do, over and over, tracing flight paths that might make ordinary air passengers sick. Their goal? Measure properties of the soot particles emitted by wildfires so they can learn how these plumes affect Earth's climate.
Correctly modeling the impact of these particles is important, the scientists say, because wildfires are increasing in both intensity and frequency, in part due to droughts brought about by increasing global temperatures and changing hydrological cycles.
"We need a better understanding of the particles emitted by these fires, including how they evolve, so we can improve our predictions of their impacts on climate, climate change, and human health," said Arthur Sedlacek, one of the intrepid smoke samplers.
Sedlacek and others at Brookhaven and collaborating institutions recently published a study in the journal Environmental Science & Technology that suggests the global climate models aren't getting the full picture.
These models base estimates of how fires impact climate on the optical properties of soot particles sampled in the immediate vicinity of a fire. As the new data show, that approach fails to account for changes in soot particles over time. Those changes, the scientists say, can dramatically influence how much sunlight the particles scatter or absorb, how they interact with water, and how likely they are to form clouds—all of which are important to how they ultimately affect Earth's climate.
"Based on these results, we should re-evaluate the use of near-source observations and laboratory experiments as the sole sources for determining how these particles are represented at the regional and global scale," Sedlacek said.
Crisscrossing smoke plumes
To gather the new data on how soot particles evolve, the scientists installed sensitive instruments designed to analyze aerosol particles on airplanes operated by the DOE and NASA.
Through two aircraft-based atmospheric science campaigns led around the world by these two agencies, they flew more than 60 research flights through wildfire plumes, back and forth at increasing distances from fires. By crisscrossing each plume repeatedly, they picked up young particles relatively close to fires, as well as particles that had evolved for hours. Other flights sampled plumes far from their sources, with ages estimated to be over 10 days.
Their main quarry was black carbon, or soot, the primary light-absorbing substance emitted by fires and the dominant particulate climate-warming agent.
"Black carbon is a great tracer to study wildfire plumes because it is inert, meaning unreactive chemically," said Brookhaven scientist Ernie Lewis, a co-author on the study. "It readily absorbs light (which is why it is black), and thus can be easily detected by our instruments."

"In addition, its only sources of removal from the atmosphere are gravity—which has little effect on these particles because of their small size—and raining out after they form cloud drops," he said.
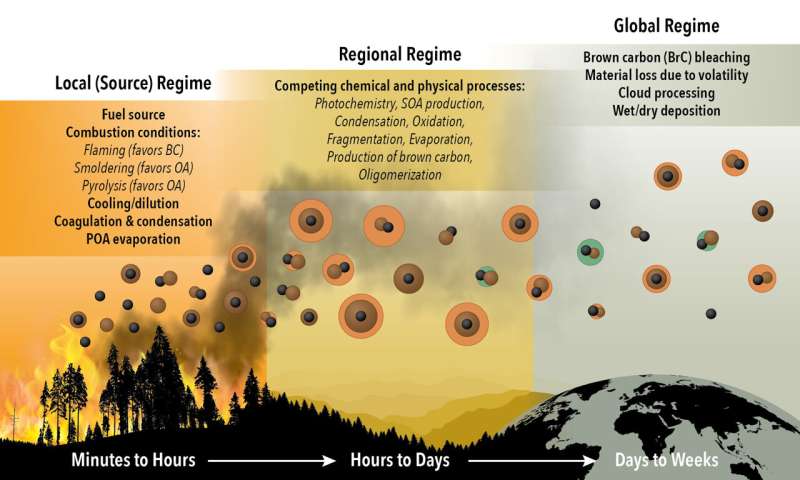
For cloud drops to form around soot, black carbon must first take on a coating of other substances. And that's where the story of simple black carbon gets a lot more complex.
Within the first hour of forming, a black carbon particle begins to accumulate a coating of organic material. This coating comes mostly from volatile organic compounds that were vaporized from the burning vegetation, which then encapsulate the black carbon "cores."
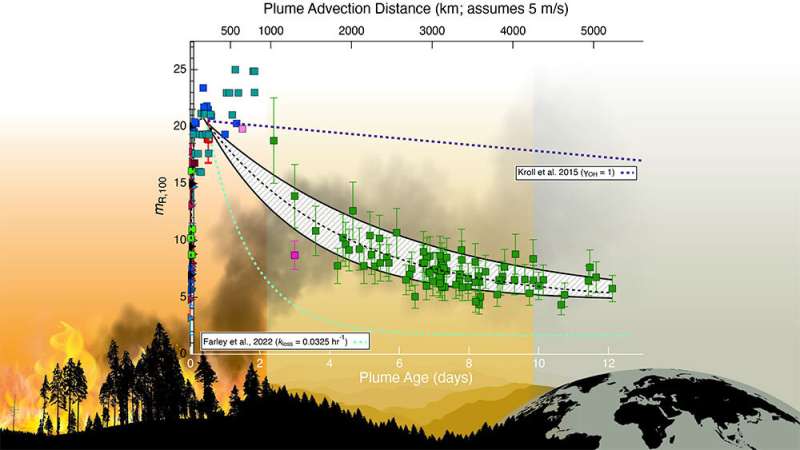
Most climate models assume that all soot particles look like these uniformly coated black carbon cores. But the data collected for this study show that the thickness of the coating material remains relatively constant for only one to two days. Then the level of coating begins decreasing slowly until only about 30 percent of it remains by about day 10 of the particle's lifecycle.
"This slow loss of coating material is not captured in today's climate models," Sedlacek said.
The data show that the particles spend a greater portion of their existence with thinning coatings—and closer to the final, 30-percent-coated stage of evolution—than any other portion of their lifecycle, which can last a couple of weeks.
Absorbing and scattering light
These changes have dramatic impacts on the particles' optical properties. For example, as the thickness of a coating on a black carbon particle decreases, the amount of light scattered by the particle decreases more rapidly than the amount of light absorbed. Since light scattering by particles generally has a net cooling effect on Earth's climate and light absorption has a net warming effect, this change in the balance alters the effect of these particles on climate.
In addition, for plumes at low elevations where clouds form, particles that have a thicker coating more readily form cloud drops. That means these particles can be removed from the plume and fall out of the atmosphere if the cloud drop becomes a raindrop. The black carbon particles remaining in the plume are therefore the ones with thinner coatings.
"Thinner coatings make these remaining plume particles relatively more light-absorbing and less scattering than the mix of particles that was in the plume before clouds formed," Lewis explained. "And once again, more absorption may trap more heat and warm Earth's climate."
Tools of the trade
The main instrument used to characterize black carbon particles is the Single Particle Soot Photometer (SP2). This instrument sends a stream of particles one-by-one through a laser beam and picks up flashes of light when the particles vaporize. It can glean information about thousands of particles per second, including their sizes and the thickness of their coatings. Here's how it works:
If a particle does not contain black carbon, it only scatters light from the laser, and the amount of light scattered can be used to determine the size of the particle.
Particles that contain black carbon aren't as simple. They scatter some light, but they also absorb light (remember, that's what makes the black carbon part of the particles black). In doing so, they rapidly heat up, triggering a series of events that occur over just a few millionths of a second.
First, the coatings evaporate, making the particles smaller, resulting in less scattered light. Next, with their coatings gone and unable to dissipate heat, the black carbon particles really heat up—to nearly 7,000 degrees Fahrenheit! That makes them vaporize and emit a flash of light. The amount of light picked up by the SP2 is directly related to how much black carbon was in the particle. Subtracting that amount from the particle size determined by the initial laser scattering gives the scientists the thickness of the coating.
This wealth of data about both the black carbon particles in the plume and the other particles can then be used to calculate how the plume as a whole interacts with sunlight and affects climate. And by making measurements of plumes at various ages, scientists can obtain better understanding of how these interactions change over the lifetimes of the plumes.
The observations made from these field campaigns are also helpful in that they draw attention to new research questions—such as how the particles in the smoke plume form cloud drops.
"Understanding the complex and intricate interactions between aerosols and clouds requires a multipronged approach where field measurements inform models, models find discrepancies necessitating targeted laboratory experiments, and where results from laboratory studies motivate new field campaigns," Sedlacek said.
More information: Arthur J. Sedlacek et al, Using the Black Carbon Particle Mixing State to Characterize the Lifecycle of Biomass Burning Aerosols, Environmental Science & Technology (2022). DOI: 10.1021/acs.est.2c03851
Journal information: Environmental Science & Technology
Provided by Brookhaven National Laboratory