March 9, 2023 feature
This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Precise transcript targeting by CRISPR-Csm complexes
Mammalian cells are inherently complex due to subcellular compartments, thereby making the process of robust transcript targeting of nucleic acids somewhat challenging in the molecular biology lab. In a recent report now published in Nature Biotechnology, David Colognori and a research team headed by Chemistry Nobel Laureate Jennifer Doudna, who discovered and expanded on the CRISPR-Cas9 technology alongside Emmanuel Charpentier, in the year 2020, incorporated a new complex to the CRISPR complex during this study.
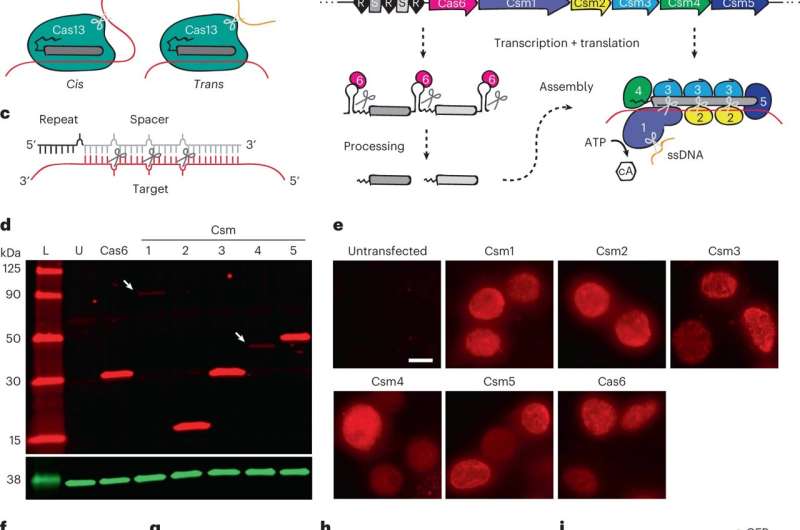
The team used the clustered regularly incorporated short palindrome repeats (CRISPR)-Csm complex; which included a protein known as Csm, a type III-A CRISPR-Cas interference complex found in the prokaryotic immune system. Thereafter, the molecular biologists accomplished surgical RNA ablation (deletion) of the nuclear and cytoplasmic transcripts via single-vector delivery. The vector-bound Steptococcus thermophilus Csm-complex provided high-efficiency RNA knockdown; a method to silence gene expression with minimal off-target impact in human cells and outperform existing genome editing technologies such as short-hairpin RNA and Cas13-mediated knockdown. By catalytically inactivating the Csm, the team achieved durable RNA binding for live-cell RNA imaging to establish the efficiency of the CRISPR-Cas effector system as RNA-targeting tools in eukaryotes.
Gene editing: From RNA interference methods to CRISPR-Cas complexes
Molecular biologists seek to alter RNA and protein levels without permanently affecting DNA; however, the task is nontrivial in basic research and in therapeutic applications. In the past, scientists had developed targeted RNA knockdown techniques in eukaryotes by using RNA interference or RNAi, whereby small interfering RNA-directed argonaute nucleases (the active part of the RNA-induced silencing complex) cleaved complementary target RNAs. However, this method led to unintended cleavage of targets carrying partially similar or complementary sequences, especially when the complementarity occurred in the nucleating "seed" region of the siRNA, while being incompatible with eukaryotic model systems.
Meanwhile, the now-widely popular clustered regularly interspaced short palindrome repeats (CRISPR) and CRISPR associated proteins (CRISPR-Cas) that form adaptive defense systems against infectious agents in prokaryotes can function as DNA or RNA nucleases that can be regulated for gene editing applications. Much like small interfering RNA, the Cas nuclease is composed of small RNAs to recognize nucleic acid targets through complementary base-pairing. In this work, Colognori and colleagues used the Csm protein as an attractive tool for RNA knockdown or gene silencing. The protein is exclusively found in prokaryotes and can be introduced to eukaryotes without the use of intersecting host regulatory pathways, allowing the research team to demonstrate the Csm system as a versatile RNA knockdown method in eukaryotes.
The all-in-one type III CRISPR-Cas system
The scientists chose the type III-A Csm complex from Streptococcus thermophilus based on a few features, such as—its existing biochemical and structural characterization in bacteria, optimal functionality, and preceding work conducted in Zebrafish embryos, and in other eukaryotic cells, including human cell cultures. The team verified the expression of individual protein components in immortalized human embryonic kidney cells and simplified the delivery of the Csm system by incorporating all components into a single vector. They used Csm to knockdown highly overexpressed and heterologous transgenes and studied the knockdown at the protein level.
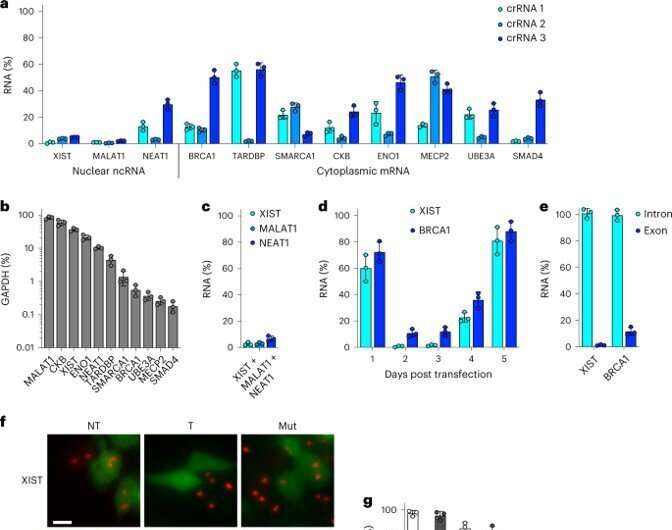
During the study, the team transfected the kidney cells with an all-in-one vector to achieve more than 90% knockdown for all RNAs of interest. The results highlighted the highly robust nature of Csm as an efficient RNA knockdown tool. The researchers verified the outcomes by performing RNA fluorescent in situ hybridization (FISH) to visualize the characteristic morphologies. The work showed how the active Csm complexes yielded similarly robust knockdown via both molecular and microbiology-related methods.
Observing RNA knockdown activity with minimal off-targets and cytotoxicity
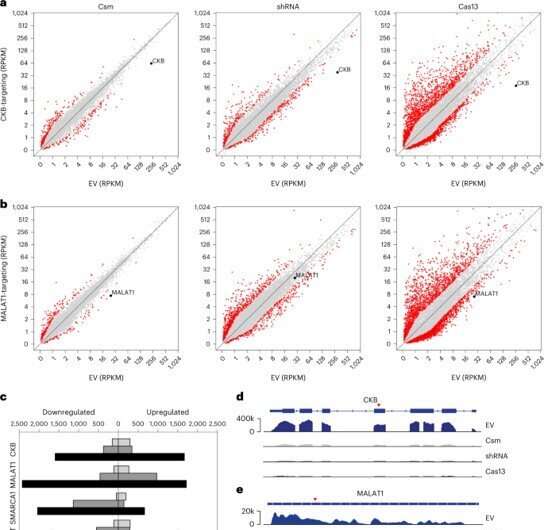
In order to assess the potential off-target effects of Csm-mediated knockdown in cells, the research team next performed RNA sequencing. They compared the efficiency of the complex with existing methods and performed knockdown for 48 hours. The team then completed fluorescence-activated cell sorting and sequencing methods with the transfected cells.
The results showed that the Csm expression did not disturb the cell environment. The researchers removed DNAse activity without affecting RNAse activity for minimal off-target effects of Csm-mediated RNA knockdown in human cells. When compared to severely cytotoxic Cas13 RNA-targeting CRISPR-Cas systems, the type III system did not display trans-cleavage activity for robust RNA knockdown and were without toxicity.
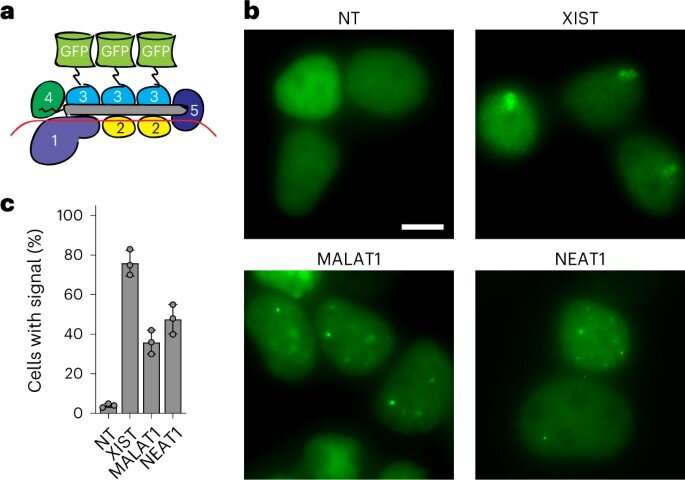
Live-cell RNA imaging without genetic manipulation
The team next sought to understand the process of tracking RNA in cells. While the process of tracking RNA in live cells is nontrivial, molecular biologists had previously accomplished the task with RNA-binding proteins such as the catalytically inactive Cas13. The researchers explored the possibility of using the Csm-complex to track RNA target in live cells by fusing green fluorescent protein (GFP) to the catalytically inactive Csm protein. Next, they transfected the kidney cell line with the Csm-GFP plasmid and assayed live-cell fluorescence microscopy after 48 hours to show that the protein could easily visualize RNA in living cells.
Outlook
In this way, David Colognori and colleagues at the Doudna Lab showed the possibility of integrating the type III-A Csm complex derived from the prokaryote S. thermophilus as a powerful tool to accomplish eukaryotic RNA knockdown and gene silencing. The team conducted RNA knockdown experiments with high efficiency and specificity when compared to existing methods and outperformed existing competition. Most notably, the process was not accompanied by detectable cytotoxicity unlike other Cas13-based methods.
The researchers facilitated competent transfection of the multicomponent CRISPR-Cas systems in to a single deliverable plasmid. The capacity to effectively bring type III systems into eukaryotes for the first time in this way has significant implications across RNA diagnostics, health screens and for synthetic biology circuits in vivo.
More information: David Colognori et al, Precise transcript targeting by CRISPR-Csm complexes, Nature Biotechnology (2023). DOI: 10.1038/s41587-022-01649-9
Abudayyeh et al, RNA targeting with CRISPR–Cas13, Nature (2017). DOI: 10.1038/nature24049
Journal information: Nature Biotechnology , Nature
© 2023 Science X Network