This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers report new mechanism in an ancient pathway of immune response

UT Southwestern biochemist Zhijian "James" Chen, Ph.D., famously identified the cGAS enzyme pathway that alerts the human immune system to disease-causing invaders like viruses. Since then, researchers have found that cGAS signaling is an ancient, conserved defense strategy stretching from bacteria to mammals. A new study from the Chen lab published in Nature reports a novel mechanism bacteria use to enhance the effectiveness of the cGAS-mediated immune response.
"This work shows that the antiviral function of the human cGAS pathway is highly conserved in bacteria and reveals a new mechanism in the arms race between bacteria and the viruses that infect them, called bacteriophages or 'phages' for short," said Dr. Chen, Professor of Molecular Biology.
Dr. Chen, also a Howard Hughes Medical Institute Investigator and Director of the Center for Inflammation Research at UTSW, received a Breakthrough Prize in Life Sciences in 2019 for his cGAS discoveries. Those advances include identifying cGAS as the enzyme that alerts the human immune system to invasion by microbes as well as several key steps in that signaling pathway. His work solved a century-old mystery and opened the door for the development of drugs designed to modulate the immune response to fight diseases such as lupus and cancer.
Long before its role as a genetic material was understood, DNA was known to activate the immune system. Dr. Chen's investigation of the cGAS signaling pathway revealed the mechanism underlying the immune response to DNA.
"The conservation of cGAS signaling across species—from bacteria through mammals—indicates the pathway's fundamental importance in the immune system," said Dr. Chen, also an investigator in the Center for the Genetics of Host Defense, member of the Harold C. Simmons Comprehensive Cancer Center, and holder of the George L. MacGregor Distinguished Chair in Biomedical Science. He explained that the Nature study reveals a mechanism that bacteria use to increase cGAS activity to overcome phage counterattacks. Viruses can only reproduce if they hijack the replication machinery of the cells they infect, so the bacteria's goal is to keep that from happening.
Viruses try to hold off the bacteria's defense long enough to replicate—a matter of minutes in some cases—and bacteria try to conquer the phages before the viruses can reproduce, the researchers said.
Dr. Chen said he hopes the work leads to a deeper understanding of human immunity at a time when infectious disease is a hot field of investigation due to the ongoing COVID-19 pandemic.
"This work is not directly related to a specific human disease but addresses a fundamental question of how bacteria fight viral infection," he said. "The study of anti-phage immune defense in bacteria led to the discovery of the CRISPR system that scientists now use for gene editing. We think ongoing studies of other pathways of anti-phage immunity—such as the cGAS regulatory pathway we uncovered in this study—may also lead to new developments in biotechnology and medicine."
The bacterial defensive pathway featuring cGAS exists along with CRISPR and other defensive systems that bacteria have developed, Dr. Chen explained. The redundancy enables bacteria to launch new anti-phage weapons when one line of defense fails.
The study represents a new direction for the lab, which previously focused on mammalian cGAS. Lead author Justin Jenson, Ph.D., recently won the University's highest honor for postdoctoral researchers—the Brown-Goldstein Award—on the basis of the experiments in the Nature study. That prize is named for UT Southwestern's first Nobel Laureates, Michael S. Brown, M.D., and Joseph L. Goldstein, M.D.
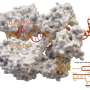
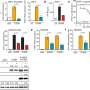
Dr. Jenson and the team identified a novel protein modification that enhances the bacterial defense against phages and works through cGAS, mirroring the enzyme's role in humans. The team then turned to the virus side of the arms race and ran a genetic screen that revealed a phage-encoded protein that fights back against the bacteria's anti-phage response pathway.
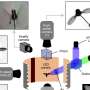
In both humans and bacteria, cGAS works in a signaling pathway to generate a second messenger, called cGAMP (cyclic GMP-AMP), to fight virus invasion. The Chen lab found that some phages, like the ones in this study, can launch a counterattack to deflect cGAMP.
"Those phages produce an agent that functions like a 'sponge' to bind and sequester cGAMP," said Dr. Chen. When successful, that counterattack nullifies the bacteria's defense by preventing cGAMP from fighting off phage infection, buying time for the phages to replicate and infect nearby bacteria.
But the battle doesn't end there. The researchers found that some bacteria have adapted to retaliate against a phage counterassault by increasing cGAS's enzyme activity, resulting in production of extra cGAMP to overcome the phage "sponge attack."
Dr. Jenson added that the battle between bacteria and phages has been evolving through billions of years, with both bacteria and phages continually mutating to create new strategies to defeat one another in an ever-escalating back and forth.
The interplay of bacterial and phage defense strategies is similar to the human response to disease-causing agents like bacteria and viruses although specific strategies vary. That's why a better understanding of the ancient bacteria-phage rivalry could lead to insights into human disease and perhaps someday to strategies to treat or even prevent them, the researchers said.
More information: Justin M. Jenson et al, Ubiquitin-like Conjugation by Bacterial cGAS Enhances Anti-phage Defence, Nature (2023). DOI: 10.1038/s41586-023-05862-7
Journal information: Nature
Provided by UT Southwestern Medical Center