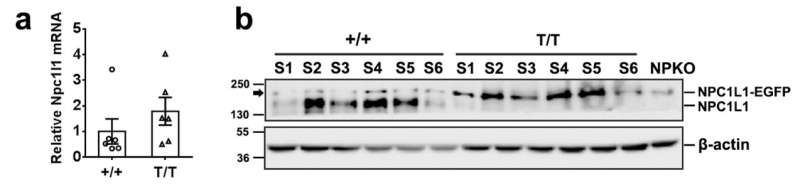
NPC1L1 mRNA and protein expressions in the small intestine of NPC1L1-EGFP mice. The small intestine was harvested from male control (+/+) and NPC1L1-EGFP transgenic mice (T/T) at the age of 8 weeks. a: The level of NPC1L1 mRNA in the S3 segments was determined by quantitative RT-PCR (n=6). b: The expression of NPC1L1 protein was detected by western blotting with anti-NPC1L1 antibody (Novus). S1: duodenum, S2-4: jejunum, S5-6: ileum. NPKO, NPC1L1 knockout. Arrow indicates the non-specific bands in the +/+ mice. Credit: Life Metabolism (2023). DOI: 10.1093/lifemeta/load011
Excessive cholesterol absorption from intestinal lumen contributes to the pathogenesis of hypercholesterolemia, which is a well-established risk factor for atherosclerotic cardiovascular disease. The absorption of intestinal cholesterol is primarily mediated by Niemann-Pick C1-like 1 (NPC1L1) protein, which is responsible for about 70% cholesterol absorption. NPC1L1-deficient mice are resistant to diet-induced hypercholesterolemia, which provides a compelling strategy for intervention the related diseases through inhibiting NPC1L1 expression or activity.
NPC1L1 protein is expressed in the brush border membrane of small intestine. The protein is extensively N-glycosylated and composed of 1332 amino acids with 13 transmembrane segments, which makes it hard to prepare an ideal antibody to analyze its behavior in vivo. Based on the studies using cell lines in vitro, it is putatively regarded that NPC1L1 mediates cholesterol absorption through clathrin-mediated endocytosis. However, this notion also has some unresolved challenge. Actually, the endocytic vesicles of NPC1L1 with cholesterol have not been demonstrated under physiological conditions, since there is no feasible tool to visualize and evaluate the endocytosis of NPC1L1 vesicles in vivo.
Using CRISPR/Cas9 gene editing technology, scientists from Naval Medical University in China generated a mouse model in which the endogenous NPC1L1 protein was tagged with enhanced green fluorescent protein (EGFP). The NPC1L1-EGFP mice enabled the researchers to fluorescently visualize and evaluate the vesicular endocytosis of NPC1L1-cago during intestinal cholesterol absorption. This study was published online in Life Metabolism.
In this study, the homozygous NPC1L1-EGFP knock-in mice were found to have normal cholesterol homeostasis on the "chow" or high-cholesterol diet conditions. The fluorescence of NPC1L1-EGFP fusion protein localized at the brush border membrane of the villus rather than the crypts in the duodenum, jejunum, and ileum, but not the colon. The pattern is consistent with the characteristics of endogenous NPC1L1 distribution in the control mice. The EGFP-positive vesicles were visualized beneath the brush border membrane as early as within 5 minutes, and peaked at 15 minutes post-oral gavage of cholesterol.
Of note, the vesicles colocalized with the early endosomal marker EEA1 and the filipin-stained free cholesterol, and cholesterol gavage triggered the accumulation of EEA1-positive vesicles beneath the brush border membrane. Pretreatment with NPC1L1 inhibitor ezetimibe inhibited the formation of these cholesterol-induced endocytic vesicles, further supporting that the vesicular endocytosis is involved in NPC1L1-mediated cholesterol absorption.
This study, for the first time, clearly demonstrates free cholesterol in NPC1L1 endocytic vesicles during intestinal cholesterol absorption under the physiological condition. It provides a feasible tool to evaluate the vesicular endocytosis of NPC1L1-cargoes as well as cholesterol absorption in vivo under pathophysiological and pharmacological conditions, and can be used in drug discovery.
In addition, given that NPC1L1 also mediates intestinal absorption of non-cholesterol sterols such as phytosterols and tocopherol, this mouse model is also beneficial for those researchers in the field of sitosterolemia and vitamin E deficiency.
More information: Xiaojing Wu et al, Fluorescent visualization and evaluation of NPC1L1-mediated vesicular endocytosis during intestinal cholesterol absorption in mice, Life Metabolism (2023). DOI: 10.1093/lifemeta/load011
Provided by Higher Education Press