This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
How protein-rich droplets form
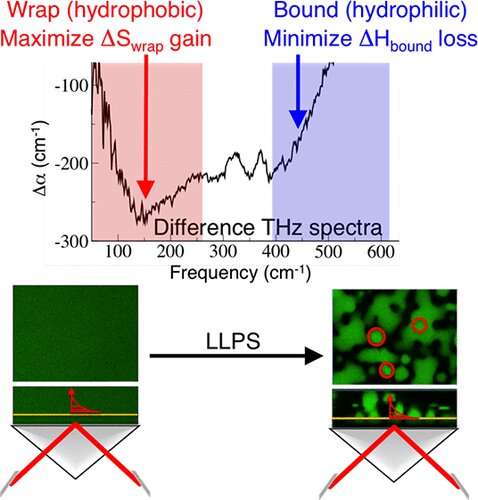
Using a new method, terahertz (THz) calorimetry, a research team from the Cluster of Excellence Ruhr Explores Solvation (RESOLV) in Bochum shed new light on the spontaneous phase separation into a protein-rich and a protein-poor phase in a solution.
It is assumed that the protein-rich droplets promote the formation of neurotoxic protein aggregates—a hot spot for neurodegenerative diseases. The researchers, led by Professor Martina Havenith, Chair of Physical Chemistry II at Ruhr University Bochum, report their findings in the Journal of Physical Chemistry Letters and Angewandte Chemie International Edition.
Molecular level and time resolution in the picosecond range
"The visionary idea in the project was to combine two powerful techniques in Physical Chemistry—laser spectroscopy and calorimetry," explains Martina Havenith.
Calorimetry measures quantities fundamental to chemical and biochemical reactions, such as heat capacity, enthalpy, and entropy. Based on well-known substance-specific parameters, it is possible to predict whether, for example, a reaction will occur spontaneously without any external input of energy or whether equilibrium conditions dominate.
Calorimetric measurements take place in a macroscopic container. The amount of heat required for a temperature change or a chemical or biochemical reaction is measured. "The limitation of this method is its limited time resolution and the amount of sample required," says Martina Havenith.
The goal of the ERC project was to overcome these limitations. This required a new approach to measure calorimetric quantities for the smallest samples with a time resolution of picoseconds, or one-millionth of one-millionth of a second at the molecular level. "However, we can not, in principle, achieve time and spatial resolution on this scale reach with these traditional concepts of heat measurements," the researcher explains. "This required a different revolutionary approach that intrinsically offers a different access."
Water plays a crucial role
The research group showed that spectroscopic fingerprints of water could be measured by their absorption in the THz range, which is linearly correlated with calorimetric quantities. This allows the researchers to track these fundamental calorimetric quantities in real time using spectroscopic and ultrafast laser spectroscopic methods, even for complex systems during a process or reaction.
In their current work, inspired by their collaboration with the research groups of Professor Konstanze Winklhofer and Professor Jörg Tatzelt at Ruhr University Bochum, they used this method for the first time to study a hot topic in Biomedical research: They investigated liquid-liquid phase separation, the spontaneous phase separation into a protein-rich and a protein-poor liquid phase.
"Using THz calorimetry, we can map the formation of these protein-rich droplets on a molecular level. Not only the proteins themselves but the water also plays a crucial role," Martina Havenith reports. "We can now follow on-line any changes in the water during the formation process with the THz camera. Based on the derived calorimetric quantities, we can give accurate predictions about the formation of phase separated droplets and the dependency on external parameters such as temperature."
More information: Simone Pezzotti et al, Liquid–Liquid Phase Separation? Ask the Water!, The Journal of Physical Chemistry Letters (2023). DOI: 10.1021/acs.jpclett.2c02697
Simone Pezzotti et al, Spectroscopic Fingerprints of Cavity Formation and Solute Insertion as a Measure of Hydration Entropic Loss and Enthalpic Gain, Angewandte Chemie International Edition (2022). DOI: 10.1002/anie.202203893
Journal information: Angewandte Chemie International Edition , Journal of Physical Chemistry Letters
Provided by Ruhr-Universitaet-Bochum





















