This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
New insights into the mechanisms of tumor growth
In many instances, the physical manifestation of cancers and the ways they are subsequently diagnosed is via a tumor, tissue masses of mutated cells and structures that grow excessively. One of the major mysteries in understanding what goes awry in cancers relates to the environments within which these structures grow, commonly known as the tumor microenvironment.
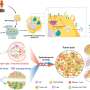
These microenvironments play a role in facilitating tumor survival, growth, and spread. Tumors can help generate their own infrastructure in the form of vasculature, immune cells, signaling molecules, and extracellular matrices (ECMs), three-dimensional networks of collagen-rich support scaffolding for a cell.
ECMs also help regulate cellular communications, and in the tumor microenvironment ECMs can be a key promoter of tumor growth by providing structural support for cancerous cells and in modulating signaling pathways that promote growth.
Now, new research led by the School of Arts & Science's Wei Guo and published in the journal Nature Cell Biology has bridged the complex structural interactions within the tumor microenvironment to the signals that trigger tumor growth. The researchers studied cancerous liver cells grown on ECMs of varying stiffness and discovered that the stiffening associated with tumor growth can initiate a cascade that increases the production of small lipid-encapsulated vesicles known as exosomes.
"Think of these exosomes as packages that each cell couriers out, and, depending on the address, they get directed to other cells," says Ravi Radhakrishnan, professor of bioengineering in the School of Engineering and Applied Science and a co-author of the paper.
"By recording the number of packages sent, the addresses on these packages, their contents, and most importantly, how they're regulated and generated, we can better understand the relationship between a patient's tumor microenvironment and their unique molecular signaling signatures, hinting at more robust personalized cancer therapies," Radhakrishnan says.
While studying exosomes in relation to tumor growth and metastasis has been well-documented in recent years, researchers have mostly focused on cataloging their characteristics rather than investigating the many processes that govern the creation and shuttling of exosomes between cells. As members of Penn's Physical Sciences Oncology Center (PSOC), Guo and Radhakrishnan have long collaborated on projects concerning tissue stiffness. For this paper, they sought to elucidate how stiffening promotes exosome trafficking in cancerous intracellular signaling.
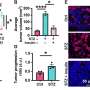
"Our lab previously found that high stiffness promotes the secretion of exosomes," says Di-Ao Liu, co-first author of the paper and a graduate student in the Guo Lab. "Now, we were able to model the stiffening processes through experiments and identify molecular pathways and protein networks that cause this, which better links ECM stiffening to cancerous signaling."
Guo's team started this process by examining the pathways that were activated when cancerous cells were grown on stiff ECMs. "The Rab family of proteins were our top candidates, as they are responsible for generating exosomes and getting them released from cells. We investigated Akt, a signaling protein that controls the Rab proteins," Guo says.
Under stiff conditions, Akt is molecularly modified, which causes it to interact with Rabin8, a protein that switches Rab family member Rab8 into its active form. Once active, Rab8 then begins its job of helping the cell release exosomes, "which further drive tumor growth," says Guo.
Later, when the researchers examined tissue samples from patients with liver cancer and tested the effects of a stiff ECM, they found that genes that code for the Notch signaling pathway, involved in cell-to-cell communications and known to promote tumor growth, were expressed more.
"The Notch pathway is implicated in liver cancer, and its activity is associated with increased liver damage," Guo says. "So, in the future, this information could be used to help clinicians diagnose cancers earlier."
"Liver cancer is a major problem in the U.S. and around the world that PSOC began to study because of the tissue stiffening," says Dennis Discher, director of PSOC. "Wei and some of his PSOC colleagues mined patient data to hypothesize and, quite remarkably, demonstrate that ECM stiffening ultimately drives tumor growth. They have demystified many aspects of the underlying mechanics which could lead to targeted drugs, but the important novel finding is the mechanosensitive triggering of bioactive release."
The work done by Wei and his team at the PSOC serves the broader goals of a program launched by the National Cancer Institute's Division of Cancer Biology known as the Physical Sciences-Oncology Network (PS-ON). Researchers working in this space seek to better understand and control cancer by applying physics, mathematics, chemistry, and engineering principles to address complex challenges in cancer research, says Eric M. Johnson Chavarria, program director for PS-ON.
Johnson Chavarria says, "This PS-ON U01 project demonstrates the importance of transdisciplinary collaboration to address outstanding questions in cancer research. I look forward to the broader impact of these physical properties and mechanistic findings between the tumor microenvironment, ECM and exosomes will have on advancing cancer research and therapies."
For future studies, the researchers want to examine the ways exosome secretions from cancerous cells affect immune cells and probe how cancers coopt fibroblasts, cells that help create connective tissue, to create more hospitable conditions in which to thrive and metastasize.
More information: Bin Wu et al, Stiff matrix induces exosome secretion to promote tumour growth, Nature Cell Biology (2023). DOI: 10.1038/s41556-023-01092-1
Journal information: Nature Cell Biology
Provided by University of Pennsylvania