Viral surface-inspired nanocarriers for improved oral insulin therapy
Clinically, type 1 diabetes (T1DM) and advanced type 2 diabetes (T2DM) patients require multiple daily insulin injections to maintain blood glucose levels, which may cause great pain and is an inconvenience to patients and can lead to hyperinsulinemia. In comparison, oral insulin therapy has high patient compliance.
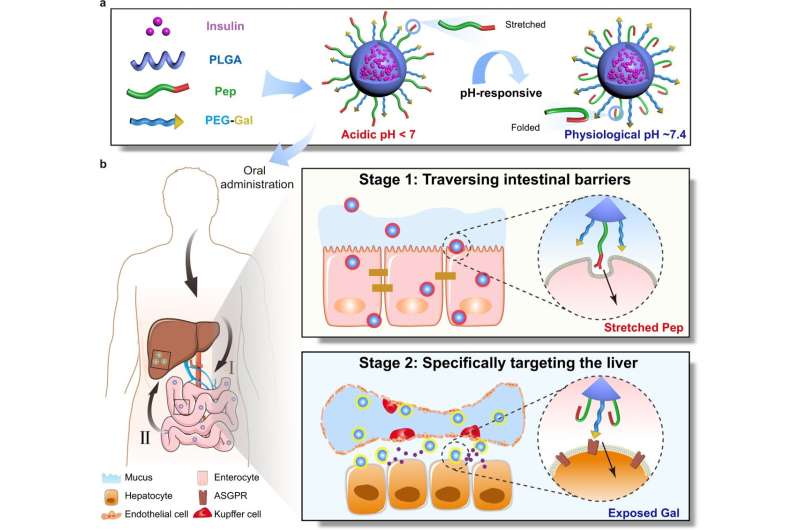
There are two bottlenecks in achieving desired oral insulin therapy. One is the gastrointestinal barrier that largely restricts the oral absorption of insulin; the other is the insufficient accumulation of insulin at target sites after absorption. Under physiological conditions, the liver is exposed to a 2–3 fold higher insulin level than the periphery and is responsible for disposing of almost a third of ingested glucose. Therefore, oral insulin delivery needs to stepwise traverse the intestinal mucosal barrier and specifically target the liver, thus simulating the biodistribution of endogenous insulin and ultimately ameliorating the utilization of blood glucose.
In a study published in Nature Communications, Prof. Gan Yong's team from Shanghai Institute of Materia Medica (SIMM) of the Chinese Academy of Sciences, in collaboration with Prof. Wei Gang from Fudan University, reported a multifunctional nanocarrier resembling viral surface for oral insulin therapy.
The researchers designed a ligand-switchable nanoparticle that was modified with a pH-responsive stretchable cell-penetrating peptide (Pep) and a liver-targeting moiety (Gal) (Pep/Gal-PNPs). Once orally administered, the acidic environments triggered the extension of Pep from the surface in a virus-like manner, enabling Pep/Gal-PNPs to traverse intestinal barriers efficiently. Subsequently, Gal got exposed by Pep folding at physiological pH, thereby allowing the specific targeting of Pep/Gal-PNPs to the liver.
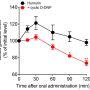
In vivo results demonstrated that Pep/Gal-PNPs exhibited effective intestinal absorption and excellent hepatic deposition (accounting for 79.1% of total absorption). Moreover, the insulin-loaded Pep/Gal-PNPs showed significant hypoglycemic effects in type 1 diabetic rats. Crucially, they increased hepatic glycogen production by 7.2 fold, improving glucose metabolism.
This study provides a novel oral insulin delivery strategy that efficiently overcomes the intestinal mucosal barriers and precisely targets the liver by mimicking the unique surface features and functions of the virus, achieving rational disposal of blood glucose in diabetes and contributing to maintaining blood glucose homeostasis.
More information: Tiantian Yang et al, Ligand-switchable nanoparticles resembling viral surface for sequential drug delivery and improved oral insulin therapy, Nature Communications (2022). DOI: 10.1038/s41467-022-34357-8
Journal information: Nature Communications
Provided by Chinese Academy of Sciences