New insights into mitochondrial nucleoid dynamics
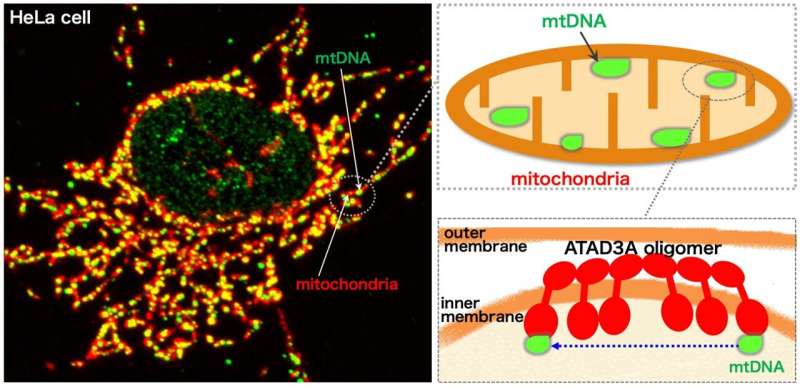
Mitochondria are important cellular structures that are vital for their role in generating energy. Mitochondria are dynamic, meaning they constantly fuse together and split apart. They contain a small amount of genetic information known as mitochondrial DNA (mtDNA). The mtDNA, organized into dot-like structures called nucleoids, also moves around inside the mitochondria.
The method of distribution of mtDNA was not entirely clear, but now a research team led by Osaka University has identified a molecule known as ATAD3A that is essential for nucleoid movement and could be a potential therapeutic candidate for mitochondrial diseases.
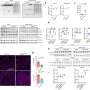
The team had previously shown that the movement of the nucleoids is linked to the fission of mitochondria. However, the mechanisms and function of this movement were unclear. The researchers therefore investigated the role of ATAD3A in nucleoid movement because of its previously established links to nucleoid formation.
In a study published recently in the Proceedings of the National Academy of Sciences, the researchers showed that ATAD3A, which is anchored to the inner mitochondrial membrane, mediated the interaction of mtDNA nucleoids (present inside the mitochondria) with factors involved in mitochondrial fission (present on the outer mitochondrial membrane).
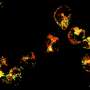
They demonstrated that ATAD3A was essential for the active movement of mtDNA nucleoids within the mitochondria—a process called nucleoid trafficking—and that nucleoids were abnormally clustered in cells that lack mitochondrial fission. Together, mitochondrial fission and nucleoid trafficking determine the size, number, and distribution of the nucleoids within the mitochondria.
The distribution of nucleoids throughout the mitochondrial network activates expression of the mtDNA and increases formation of the respiratory chain complex, which is a group of several proteins essential for energy production within cells, and the correct distribution of the mtDNA nucleoids is key for efficient energy production.
"Regulation of nucleoid dynamics is crucial for the maintenance of respiratory chain complexes on the mitochondrial inner membrane," explains lead author Takaya Ishihara, "and this is the first report of the role of ATAD3A in mitochondrial nucleoid dynamics."
The development of techniques to alter mtDNA movement may help regulate mitochondrial function in the future. "Nucleoid trafficking may represent a new therapeutic target to prevent mitochondrial dysfunction in various human diseases," explains senior author Naotada Ishihara.
Therefore, not only does this work increase our knowledge of the regulatory processes in mitochondria but it also provides scope for developing future therapies against abnormal mitochondrial functioning.
More information: Takaya Ishihara et al, Mitochondrial nucleoid trafficking regulated by the inner-membrane AAA-ATPase ATAD3A modulates respiratory complex formation, Proceedings of the National Academy of Sciences (2022). DOI: 10.1073/pnas.2210730119
Journal information: Proceedings of the National Academy of Sciences
Provided by Osaka University