A new catalyst to slow down global warming

Russian scientists have developed a new, highly efficient catalyst for carbon dioxide industrial processing that makes the process simple and inexpensive. Scientists from MISIS University, Lomonosov Moscow State University and Zelinsky Institute of Organic Chemistry took part in the study. The results have been published in Materials.
One of the promising ways to process greenhouse gas–related carbon dioxide is through the reaction of its interaction with hydrogen. According to the scientists, the valuable products of this reaction can be the synthesis gas, various hydrocarbons and alcohols widely used in the chemical industry. Scientific teams around the world are struggling to find sufficiently efficient and durable catalysts (compounds that accelerate the course of that chemical reaction) that will allow the scaling up of carbon dioxide processing for a green economy.
Scientists from MISIS University together with their colleagues from the Lomonosov Moscow State University and the Zelinsky Institute of Organic Chemistry have developed a new simplified method of producing industrial cobalt-nickel catalysts for carbon dioxide processing.
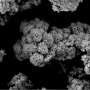
"Our catalysts are a bulk alloy with a porous surface and nanoscale grains that form foamy high-activity particles. Due to this structure and the synergistic interaction of Co with Ni, the catalysts are characterized by a more intense interaction with CO2 molecules and high stability, compared to existing analogs (active element dispersed on a ceramic carrier)," explained Sergey Roslyakov, Senior Researcher at NUST MISIS.
The scientists focused on three problems: exploring the possibilities of full utilization of carbon dioxide (which enhances the greenhouse effect on the planet), as well as simplifying the production of effective catalysts and creating catalysts based on available raw materials.
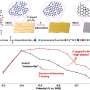
"Our work is distinguished by the rapid and simple synthesis of material via combustion of reactive sol-gels. In our approach, it is enough to apply insignificant energy to heat a small volume of the sample, up to one cubic millimeter in size, and then the synthesis proceeds in a self-sustaining mode without additional energy costs," Roslyakov said.
The use of non-standard synthesis methods has significantly reduced energy and resource costs in the production and use of the catalysts. According to the authors, cobalt contributes to the formation of a porous sponge-like microstructure of the catalyst and also triples the catalytic properties of nickel.
Since the entire volume of the catalyst consists of a metal alloy, it has a much higher thermal conductivity compared to ceramic carriers. As they explain, this significantly increases the stability of the material during long-term use.
"We have simplified the method of materials preparation, avoiding long and non-trivial stages of melting, spraying, cleaning, application of active components on the structure-forming carrier and others. Despite the simplified synthesis process and the composition of the catalyst, we have obtained a competitive technology for the catalytic conversion of carbon dioxide," Roslyakov added.
In the future, the scientific team intends to continue the search for new effective and stable catalysts.
More information: Nikolay Evdokimenko et al, Sponge-like CoNi Catalysts Synthesized by Combustion of Reactive Solutions: Stability and Performance for CO2 Hydrogenation, Materials (2022). DOI: 10.3390/ma15155129